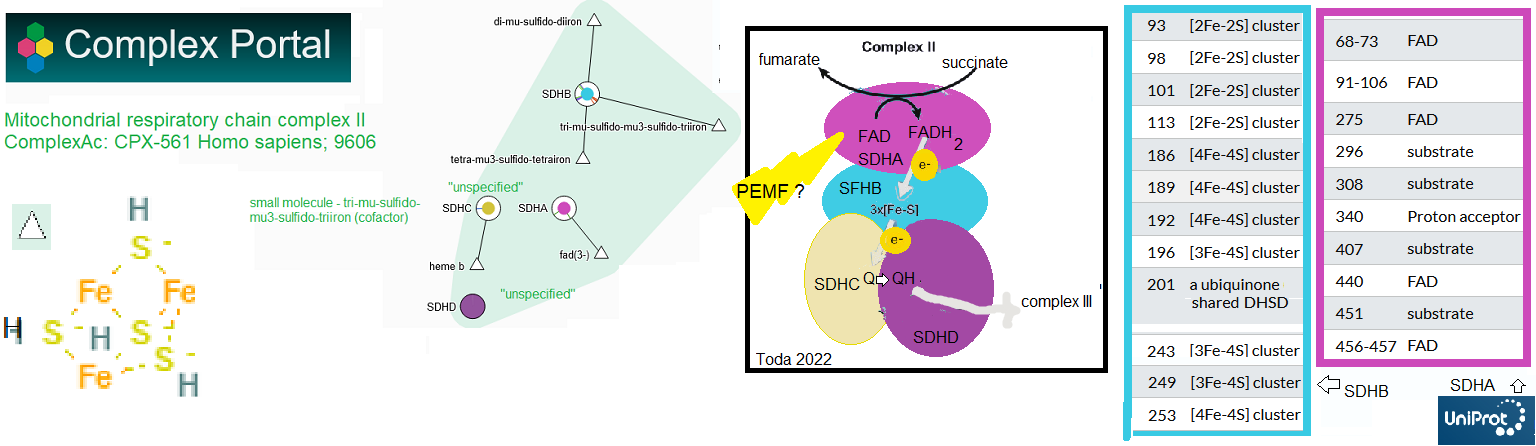
According to the Complex Portal Complex II aka succinate dehydrogenase (SDH: SDHA-D), is made up of four protein subunits along with iron-sulfur (FeS) centers. Complex Portal links t the Swiss site UniProt.org. UniProt annotates the amino acid sites of proteins that interact with substrates and cofactors. Toda and coworkers 23023 investigated the effect of optimized extremely low frequency weak magnetic fields (Opti-ELF-WMF) on the mitochondria of mouse livers and in cultured hepatocytes. The flavin adenine dinucleotide (FAD) and succinate binding SDHA subunit was found to be crucial for the effects of Opti-ELF-WMF. The effect was deemed to be generation of reactive oxygen species. This post also acknowledges the extensive number of lysines reported to be acetylated and the report of Han 2010 that the deacetylase sirtuin 3 modulates the activity of mouse SDHA. These observations suggest dietary supplement ways of making PEMF work better.
The protocol in brief [1]
This study out of Japan used a device called the Opti-ELF-WMF,
- weak field strength of 10 µT. 4 ms pulse width with increasing pulse frequencies of 1, 2, 3, 4, 5, 6, 7, and 8 Hz in 8 s
- a common strain of laboratory mouse, and a hepatocyte cell line.
- The mice were exposed in their cages. It was not quite evident if this was 24/7 or what. Open field tests were also mentioned but not reported in the main publication.

Mitophagy overview. (1) Membrane potential is lost and membranes start leaking. (2) Pink and Parkin move in sensing the decreased membrane potential causing phagosome formation (3) that fuses with the lysosome (4) which degrades mitochondria proteins (5)
AbCam has a nice overview of step 1. PINK is a protein kinase that can activate/phosphorylate itself. Activated PINK may recruit Parkin, a ubiquitin ligase to the surface of the mitochondria. Parkin attaches ubiquitin strings to mitochondria surface proteins that serve as an “eat me” signal.
summary of figures
- Exposure to Opti-ELF-WMF for 4 weeks increased mitochondrial electron transport chain (ETC) activities in the mouse liver. In addition to an increase in oxygen consumption, specific activity of complex IV was increased slightly with statistical significance.
- In cultured hepatocytes, in just a few hours, Opti-ELF-WMF deceased reactive oxygen species ever so slightly followed by an increase over the course of several hours. Mitochondria mass and membrane potential decreased in the 3-6 hour time frame. (Step 1 of mitophagy.) Opti-ELF-WMF decreased mitochondrial mass to 70% and the mitochondrial electron transport chain (ETC) complex II activity to 88%.
- By 3 hours there was a decrease in the relative amounts of many mitochondrial proteins themselves. This could be due to decreased production or increased degradation. By 12 hours there was no difference between control and Opti-ELF-WMF.
- By two hours there was an increase in lysosome targeting light chain 3 (LC3). A significant increase in a mitophagy dye was seen at 2 hr with a peak at 150 min. Parkin was also observed to translocate from the cytosol to the mitochondria. Lysosome and mitochondria specific dyes were observed to merge. The lack of PINK1 negates the effect of ELF-WMF. The suppression of complex II of the ETC was concluded to induce mitophagy by translocating Parkin and PINK1 to the mitochondria and by recruiting LC3-II.
- PGC-1α is a key co-transcription factor in mitochondria upkeep. It partners with transcription factors PPARα and TFAM for mitochondrial biogenesis and metabolism. The Opti-ELF PEMF treatment increased the expression of PGC-1α and aPPARα but not mitochondrial genome transcription factor TFAM suggesting that PGC-1α-mediated mitochondrial biogenesis was activated after mitophagy to rejuvenate mitochondria. Protein ubiquitination was also increased. The fatty acid and PEMF influenced transcription factor SREBP1 was not examined in this study, SREBP was covered in the PEMF and fatty liver post. This Zhai 2023 does not include involvement of SDHA of complex II. The link of mitophagy to PGC-1α-mediated mitochondrial biogenesis to rejuvenate mitochondria.
- Mice were exposed again to Opti-ELF-WMF again, this time for only 8 minutes instead of four weeks. ETC complex II was reduced to 88% while complexes I, III, and and IV were unaffected by these short exposures. Complex II was then dissected into subunits: SDHA, SDHB, SDHC, and SDHD. Chemical inhibition of electron flux through the mitochondrial ETC complex II nullifies the effect of ELF-WMF. These authors established SDHA as the target of AOpti-ELF-WMF. [6]
The Franco-Obregón model revisited
The Franco-Obregón model is less dependent on SDHA being the antenna for the effects of PEMF on the liver.
Post translational modifications of SDHA
The Toda study [6] may offer even more ways to rejuvenate mitochondria. This site has discussed the sirtuins, enzymes that remove regulatory acetyl groups from lysine side chains. A proteomics study wished to establish targets in a Sirt3 knock out mouse. Not only is SDHA heavily acetylated, its acetylation inhibits activity. [7] Some of the lysines in the mouse sequence include K179, K485, K498, and K538, which are conserved in the human sequence. This figure was the mouse HDHA entry from UniProt. All sources reviewed on this post point to PEMF generation of reactive oxygen species. Toda 2024 [6] made an excellent point for the SDHA subunit of mitochondria complex II as an antenna. Cimen 2010 [7] made an excellent case for Sirt 3 regulating acetylation of SDHA. Could this confound ROS from PEMF?

None of this is simple. SDHA seems to be an excellent antenna for how transcranial PEMF might affect brain function, particularly in the mitochondria. With a few phosphorylation and lycine succinylation sites and massive amounts of potential acetylation sites, it is not that simple!
The ion cyclotron resonance model reminds us that a magnetic field can set in motion movement of ions. [7] While the authors had a cartoon of a voltage gated calcium channel in their cartoon, this ICR model has nothing to do with opening of the voltage gate but could have something to do with flow of ions in the Helmholz layer of ions on the surface of the cell to the channel opening, and/or through the channel cavity once the gate is open. This model could also follow the flow of an electron through complex II, which interacts with FAD in addition to having heme and Fe-S centers. The Franco-Obregeon Lab image has been supplemented with an image from Research Gate and a very nice Fe-S center cartoon from Dr Sumbul Fatma’s slideshare.
Magnetic field antenna in complex II

The image to the left is an aid to viewing Yap 2019 [5] Figure 1 Graphical abstract. Schematic representation of how magnetic fields activate a TRPC1-mitochondrial axis upstream of calcineurin/NFAT and PCAF genetic and epi genetic pathways, promoting myogenesis and mitochondria genesis. The image to the left illustrates the potential of Fe-S centers in complex II to act as antenna
References
- Toda T, Ito M, Takeda JI, Masuda A, Mino H, Hattori N, Mohri K, Ohno K. Extremely low-frequency pulses of faint magnetic field induce mitophagy to rejuvenate mitochondria. Commun Biol. 2022 May 12;5(1):453. PMC free paper
- Cimen H, Han MJ, Yang Y, Tong Q, Koc H, Koc EC. Regulation of succinate dehydrogenase activity by SIRT3 in mammalian mitochondria. Biochemistry. 2010 Jan 19;49(2):304-11. PMC free paper
Leave a Reply