What happens when to a molecule when it absorbs visible light? A 1999 Scientific American post explored the question of why a brick wall heats up when it absorbs light. Two explanations were given to this seemingly simple lay question:
- Tom Zepf of the physics department at Creighton University claims that the molecular structure of brick resonates well with infrared radiation from the sun. This makes sense from an intuitive level. Water heats up in a microwave oven whereas and empty plastic cup does not.
- Scott M. Auerbach, a theoretical chemist at the University of Massachusetts at Amherst claims that absorption of elctromagnetic radiation excites electrons to higher molecular orbitals to an electronically excited state. Instead of emitting a photon equivalent to the energy difference between the excited and ground state in a process that many of us know as fluorescence, the excited electron collides with a neighboring molecule causing it to vibrate more. Auerbach calls this a “vibrationally excited state.” A quarter of a century later LibreTexts reiterate this truth of decay of the electronically excited state to a vibrationally excited state. This explanation resonates with Healtline’s definition of cold laser therapy, i.e. use of a low intensity laser that does not heat up the body.
What happens when light strikes an object?
- It is reflected away from the surface
- It passes through the object
- It excites valence electrons that return to the ground state in a process we call fluorescence that is immediate and phosphorescence in which the shorter wavelength emission and return to the ground state is delayed.
- It excites valence electrons that return to the ground state by colliding with neighboring atoms whose vibrational/thermal energy is increased
Possibility #4 is particularly intriguing because the rate of any reaction is dependent on temperature. Might localized increases in thermal energy increase catalysis or decrease he energy of activation? Soluble guanylyl cyclase offers a particularly intriguing model to study this basic question. sGC has strong absorbance at in the blue region and nm in the green region of the visible spectrum. Weak absorbance is seen in the nm red region. One may argue that the red absorbance matters more because shorter wavelengths scatter more.
Is soluble guanylyl cyclase light sensitive [1,2] ?
A soluble fraction from morning glory, P nil, had SNP and YC-1 activated guanylyl clyclase activity. White and blue light activated this activity while red light and darkness inhibited GC activity. [1] In isolated rat occipital arteries1,460 nm infrared caused a cGMP mediated vasorelaxation that was presumably due to liberation of nitric oxide from nitrosothiols. [2] A previous study demonstrated photodecomposition of nitrosoglutathione at 550 nm in addition to the previously described 350 nm degradation. 2GSNO ➔ GSSG + 2-NO [3]

Difficult to find absorption spectra of SNOs
The unanswered question is whether blue light at 450 nm would be just as good as 550 nm red light when it comes to the photo decomposition of SNOs. It should be pointed out that the 1995 Singh study ued Rose Bengal to enhance the degradation of nitrosogluathione. [3]
Soluble guanylyl cyclase, the absorption spectrum [4]
heterodimeric full-length and N-terminal fragments of Manduca sexta sGC in Escherichia coli and was found to behave like its mammalian counterpart in being stimulated 170-fold stimulation by NO and sensitivity to compound YC-1, a compound that reduces the NO and CO off-rates.
Binding of NO leads to a transient six-coordinate intermediate, followed by release of the proximal histidine to yield a five-coordinate nitrosyl complex (k6-5 = 12.8 s-1).
UV-visible absorption maxima and extinction coefficients for msGC-NT2
| Ligand | Soret | β | α |
|---|---|---|---|
| 433 (149 ± 2)a | 557 (20) | NDb | |
| CO | 425 (221) | 542 (21) | 572 (21) |
| NO | 400 (127) | 544 (19) | 574 (20) |
in parentheses (mm–1 cm–1). The extinction coefficient for the unliganded Soret band was determined from direct measurement of heme content, with the error estimated from the standard deviation of four measurements. All other values were derived from the ratio of the absorption maximum to that of the unliganded Soret band, ND, not distinct. Note that the 400-433 Soret bands have easily 5-10 x the extinction coeffcients of the Q-bands. Could the heme group catalyze the photodecomposition of any SNO near it? This post will cover inhibitory SNO in the catalytic region of sGC rather than the NO binding region.
Fig 2. SDS-PAGE and UV-visible absorption spectra for msGC-NT2. The authors demonsrate that they are dealing with pure protein and that NO and CO bound make a difference in the wavelength of light that is absorbed, mostly in the blue region of the spectrum.
NO binding induced the breakage of the proximal Fe-His bond, producing 5-coordinate NO-heme, with ~400 nm Soret absorption bands
Retinal expression of soluble guanylyl clyclase? [5]
This post being a continuation of the Green Light for Pain post, it might be useful to review some of the cell types covered in this post. This is an acknowledgement that In the mammalian retina membrane bound guanylyl cyclases tend to be controlled by calcium sensing proteins signalling. The Weinberg Laboratory at the University of North Carolina Chapel Hill conducted an elaborate immunohistochemistry study that mapped soluble GC in cell types of the retina.
- outer and inner segments of photoreceptor cells
- preference for cone rather than rod bipolar cells
- in GABAergic amacrine cells
- ganglion cells
- probably not in Müller glial cells
- tendency for co-localization with neuronal nitric oxide synthase, particularly in amacrine cells
As for red light therapy increased blood flow, blue, green, and other shorter wavelengths are not going to penetrate the skin as well.
Nitrosothiols, inhibitors of sGC activity [6]
Dr Annie Beuve only stated that experiments on sGC was pretreated with 50 μM GSNO in the dark for 10 min. “The SNO bonds are light sensitive, which probably explains why desensitization of sGC was not observed before, when GSNO was used as a NO donor.” Can the heme group of sGC act as a photo catalyst of protein bound SNO just as Rose Bengal catalyzes the decomposition of GSNO {3]?
Figure 1, many validations of technologies used
These authors used the biotin switch to detect SNOs. Endogenous sGC is S-nitrosylated and loses NO responsiveness in primary aortic SMC treated with CSNO.
- (A) (Left) Western blot with anti-α and anti-β antibodies showing that sGC from the cytosols of CSNO-treated cells is immunoprecipitated by anti-SNO antibodies whereas no sGC is pulled down from l-Cys-treated cells. (Right) Western blot analysis with anti-sGC of a biotin switch assay followed by avidin purification, confirming that sGC is S-nitrosylated by CSNO treatment. Input corresponds to 10% of the 200-μg precleared cytosols. One hundred micrograms of cytosols was used for the biotin/avidin assay. IgG did not immunoprecipitate sGC from CSNO-treated cells, indicating specificity of anti-SNO. Semipurified rat sGC (300 ng) was used as marker: α- and β-subunits are 80 and 72 kDa, respectively.
- (B) The same biotin-avidin assay as in A showing that sGC is S-nitrosylated in HUVEC treated with VEGF for 1 h at 10 ng/ml.
- (C) Biotin-avidin assay followed by Western blot with anti-sGC showing that sGC is S-nitrosylated in aorta treated with Ach for 1 h (100 μM). These blots (A–C) are each representative of three independent experiments.
- (D) Basal and NO-stimulated sGC activity of the cytosols prepared in A showing that the sGC of cytosols from CSNO-treated cells loses responsiveness to NO; basal and NO-stimulated activity (at 1 and 100 μM of the NO-donor SNAP) were not significantly different, in contrast to NO-stimulated activity of cytosols from l-Cys treatment. These experiments were repeated three times with each measurement done in duplicate and expressed as mean ± SE (*, P < 0.05).
Fig 1 data argue for the integrity of the protocols and that SNO is a physiological event, not some artifact.
Figures 2-3 SNO turns sGC activity down
Fig 2 S-nitrosylation and desensitization of sGC is a function of time and concentration of CSNO and is reduced with NAC pretreatment. The following is a direct quote of the figure legend that does an excellent job of describing what was done.
- “(2A) Western blot with anti-α and anti-β of a biotin-avidin assay showing that S-nitrosylation of sGC in SMC increases with time and concentration of GSNO. The input (10% of the cytosol) indicated similar sGC levels in each sample.
- (2B) Basal and NO-stimulated activity of the various cytosols from A showed that the decrease in NO-stimulated sGC activity was a function of the concentration and time of CSNO treatment. The small increase in basal activity with 1 mM CNSO was not significant. *, P < 0.05 vs. l-Cys.
- (2C) (Left) Western blot with anti-sGC showing that less sGC is precipitated with anti-SNO from the cytosols of cells treated with NAC and CSNO than from cells treated with CSNO only, whereas none is pulled down with l-Cys or l-Cys + NAC treatment or with IgG. (Right) Western blot showing that more sGC is detected in fractions not precipitated by anti-SNO, hence not S-nitrosylated, of cells pretreated with NAC + CSNO than with CSNO alone; α and β are indicated by arrows (the unspecific band between α and β has the same intensity in all samples). Two hundred micrograms of cytosols was used in the experiments and repeated twice with similar results.
- (2D) Basal and NO-stimulated sGC activity of treated cytosols from C. NO-stimulated activity was inhibited by >50% with 250 μM CSNO but pretreatment with NAC blocks the CSNO-dependent desensitization. Experiments were repeated three times and are expressed as mean ± SEM. *, P < 0.05 CSNO vs. CSNO + NAC. Note that some of the differences can be explained by batch variations of primary aortic SMC; activities are not normalized.”
Fig. 3.The NO-stimulated activity of semipurified sGC is reduced by pretreatment with GSNO and correlates with its S-nitrosylation.
- “(3A) NO-stimulated activity was reduced by ≈50% with 50 μM GSNO pretreatment compared with GSH. Measurements were made after 100-fold dilution of the sGC + GSNO or GSH mixture and in the presence of 100 μM SNAP as an NO donor to the heme group. This experiment was repeated three times and with two independent preparations of sGC.
- (3B) In parallel, sGC was S-nitrosylated by 50 μM GSNO, as shown by a biotin switch assay followed by immunoblot analysis with antibiotin and anti-sGC. Note that the amount of sGC seems to be about the same but the amount of NO adducted thiols is next to nil in the presence of GSH.”
We still do not know which thiols are modified at this point.
Figures 4-5, cysteines to nonreactive alanines
These figures have two stories. Figure 4 shows mass spctrometry results which identify the cysteines that are modified by the “biotin switch” disulfide bond. Figure 5 analyzes a system in which a expression vector encoding the α1- and β1-subunits of rat sGC with the α1C243A and β1C122A mutations were introduced to COS-7 cells. Figure 4 shows a representative spectrum with masses (M/z) corresponding to tryptic fragments with the break up of the disulfide biotin probe and protein cysteimes.
Fig. 5.Mutational analysis of the S-nitrosylated Cys in sGC indicates that α1C243 and β1C122 are involved in sGC desensitization.
Mutagenesis of some of these residues to alanine removes the ability of GSNO to inhibit guanylyl cylase activity.
More sGC SNOs from the Bueve Lab [6]
This time the starting material was rat prenatal cardiac myocytes. The authors captures a much larger array of nitrosated thiols with the biotin switch protocol. They seemed quite focused on the nitrosated thiols in the catalytic domain for obvious reasons.
Back to what happens when a object absorbs light
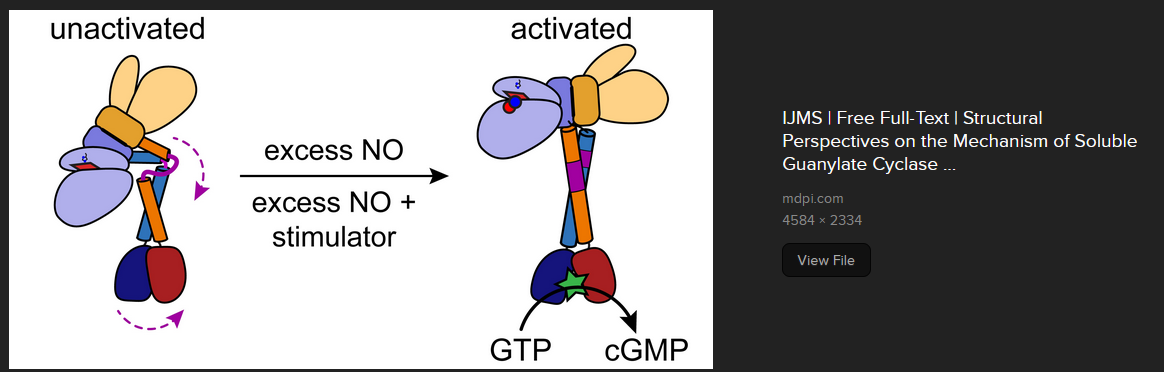
There is Cys 74 in the human sequence, whose equivalent in the rat sequence, is not nitrosated. The rscb.org sources of the activated and un-ligated structures are shown. What if there were nitrosothiols close to the heme group that are never nitrosated in the light but may be nitrosated in the dark?

Cys78 is considered to be part of the heme binding pocket in both forms shown here. Cys78 forms a hydrogen bond with the carbonyl group of Phe74. The only problem with this scenario is that Light at 350 and 550 could theoretically turn sGC on. This would not work in the retina because light could result in the production of more cGMP. On the other hand it could explain the increase in blood flow in response to red light therapy.
The Cys174 in the β sub unit is interesting. It is sort of close to the heme group. Ironically, Cys122 seems to be closer, but was not found to be nitrosated in the second study [6], but was nitrosated in the 2007 study [5]!

The post is not even addressing the role of thioredoxins and other enzymes removing NO adducts in the catalytic domain of sGC.
References
- Szmidt-Jaworska A, Jaworski K, Kopcewicz J. Effect of light on soluble guanylyl cyclase activity in Pharbitis nil seedlings. J Photochem Photobiol B. 2008 Oct 16;93(1):9-15.
- Lewis THJ, Zhuo J, McClellan JX, Getsy PM, Ryan RM, Jenkins MJ, Lewis SJ. Infrared light elicits endothelium-dependent vasodilation in isolated occipital arteries of the rat via soluble guanylyl cyclase-dependent mechanisms. Front Physiol. 2023 Aug 16;14:1219998. PMC free article
- Singh RJ, Hogg N, Joseph J, Kalyanaraman B. Photosensitized decomposition of S-nitrosothiols and 2-methyl-2-nitrosopropane. Possible use for site-directed nitric oxide production. FEBS Lett. 1995 Feb 20;360(1):47-51. free paper
- Hu X, Murata LB, Weichsel A, Brailey JL, Roberts SA, Nighorn A, Montfort WR. Allostery in recombinant soluble guanylyl cyclase from Manduca sexta. J Biol Chem. 2008 Jul 25;283(30):20968-77. PMC free article
- Ding JD, Weinberg RJ. Distribution of soluble guanylyl cyclase in rat retina. J Comp Neurol. 2007 Feb 1;500(4):734-45. doi: 10.1002/cne.21206. Erratum in: J Comp Neurol. 2007 May 1;502(1):171. Corrected and republished in: J Comp Neurol. 2007 May 1;502(1):734-45. Sci-Hub free article
- Sayed N, Baskaran P, Ma X, van den Akker F, Beuve A. Desensitization of soluble guanylyl cyclase, the NO receptor, by S-nitrosylation. Proc Natl Acad Sci U S A. 2007 Jul 24;104(30):12312-7.PMC free article
- Beuve A, Wu C, Cui C, Liu T, Jain MR, Huang C, Yan L, Kholodovych V, Li H. Identification of novel S-nitrosation sites in soluble guanylyl cyclase, the nitric oxide receptor. J Proteomics. 2016 Apr 14;138:40-7. PMC free article
Leave a Reply to PEMF and hemoglobin – BDLbiochemCancel reply