PEMF is said to increase oxygen delivery to our bodies. Just exactly what does this mean? This post presents a hypothesis as to the molecular and electromagnetic mechanism as to how this occurs.
O2 binding to hemoglobin basics
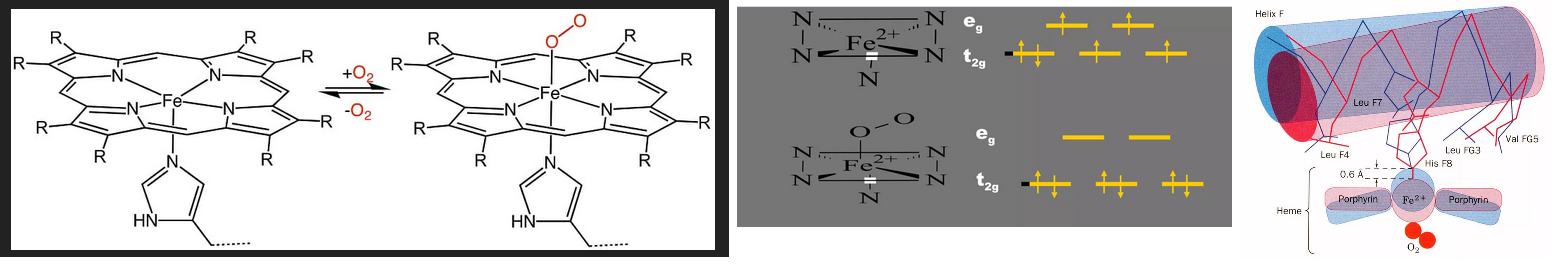
Before getting to the featured papers we’ve got to do a quick study on the oxygen binding of hemoblobin. Teach Me Physiology does a good job of this. the percentage of oxygen bound to haemoglobin is related to the partial pressure of oxygen (pO2) at a given site. When oxyhaemoglobin reaches a tissue with a low pO2 (e.g. skeletal muscle), it will dissociate into oxygen and haemoglobin, resulting in an increase in local pO2. When it reaches a tissue that has a high pO2 (e.g. in the pulmonary circulation), haemoglobin will continue to take up more oxygen, resulting in a lowered pO2. Slide Share has a good piece on hemoglobin biochemistry. The featured image is slides 26 and 43 of 114 slides. 2,3-DPG/BPG destabilizes the deoxy T-form by creating more contacts for oxygen binding to Hb. O2 binding to Hb causes dissociation of 2-DPG because the cavity gets too small also favoring the R relaxed form. This particular slide share did not explain the spin paired and unpaired electrons.
Theoretical in silico predictions
A search to discover what this means netted a Turkish paper describing magnetic nature of O2 binding to heme iron. [1] These authors used computer simulations to show that local magnetic moments can develop in the porphyrin layer with antiferromagnetic coupling to the Fe moment.
Figure 2. Magnetic-moment density.
“Illustration of the magnetic-moment density M(r) for (a) the deoxy and (b) oxy-heme clusters at T=150 K (-123oC). Here, the red (blue) color indicates the atomic orbitals which have magnetic moments pointing up (down). The magnitude of M(r) at an atomic site is proportional to the volume of the bubble at that site. The calculation of M(r) is described in the Supplementary Information.” Note the directionality of Fe d shell unpaired electron (red) and the double bond (π) electrons (blue) in the heme ring.
Here are some links of things to look at in figures that may or may not be copyrighted.
Figure 2 Mayda 2020 Magnetic-moment density. Illustration of the magnetic-moment density M(r) for (a) the deoxy and (b) oxy-heme clusters at T = 150 K. Note the position of the Fe relative to the heme ring. Figure 4 (a) Illustration of the spin polarizations for the Fe(3d), and the bonding π and the antibonding π⁎1 and π⁎2 host states in an applied magnetic field Bapp for deoxy-heme. For Bapp pointing up, the total Fe(3d) spin gets polarized in the down direction, while the spin of the nearly half-filled π⁎1 state gets polarized parallel to the field. The π⁎2 state is nearly empty. The potential of PEMF antenna in the pi an d-shell electrons is interesting.
Figure 4
The authors introduced the concept of left and right polarized UV light. Biochemists tend to use the UV range of the visible spectrum to measure the concentration of compounds that have a lot of double bonds.
“(a) Illustration of the spin polarizations for the Fe(3d), and the bonding π and the antibonding π1⁎ and π2⁎ host states in an applied magnetic field Bapp for deoxy-heme. For Bapp pointing up, the total Fe(3d) spin gets polarized in the down direction, while the spin of the nearly half-filled π1⁎ state gets polarized parallel to the field. The π2⁎ state is nearly empty. From the DFT+QMC results we know that the π states are located about 3 eV below the Fermi level, and the widths of the π ⁎ 1 and π2⁎ states are about 0.3 eV.
(b) Feynman diagram representing the absorption of LCP light in the optical transition π → π1⁎. Here, an up-spin electron in a π state makes a transition to the π1⁎ state becoming down-spin by absorbing LCP light (denoted by the wavy line).
In (c), the π → π2⁎ transition is shown for the absorption of RCP light, where a down-spin π electron makes a transition to the π2⁎ state becoming up-spin. Here, the red dot is the effective vertex for the optical transition. Even though there is spin-orbit interaction only at the Fe(3d) orbitals, the π states can gain an effective spin-orbit coupling through anti-ferromagnetic coupling and hybridization with the Fe(3d) orbitals, which are discussed in the Supplementary Information.”
This post will not attempt to understand the physics of electron spin coupling between d shell and π electrons. What we know about this physics predicts is that heme oxygen binding might be influenced by magnetic fields in vivo. Here is in vitro proof. When reading the next section, bare in mind the rather complex regulation of O2 binding to the hemoglobin tetramer, not just one of four heme groups.
experiments: pulsed radio waves and astatic magnetic field
It is rather difficult to communicate in this post just how exactingly the Muehsam study was in controlling conditions of exposure and the potential of heat generation, orientation side effects, and so on. Here are a few bullet points of what they did.
- All hemoglobin samples were exposed to the ambient magnetic field, which was measured using a digital Gauss/Tesla meter (model 7010, F.W. Bell, USA) to be 40.5±2 µT, 59 degrees from horizontal (vertical component = 34.7±2 µT, horizontal component = 21.0±2 µT).
- The FDA approved pulsed radio frequency PRF signal consisted of a 27.12 MHz sinusoidal carrier through pulse modulation in 4 ms bursts, repeating at 5 Hz and peak magnetic field amplitude of 10±1 µT. (Roma3, Ivivi Health Sciences, San Francisco, CA, USA) The authors went into great detail as to the clinically approved use for pain management in humans and animals, the exact orientation of the hemoglobin samples, origins of the devices, and so on.
- Static magnetic field SMF exposure was delivered using circular permanent ceramic magnets constructed for therapeutic applications (Magnetherapy, West Palm Beach, FL, USA), and composed of compacted and sintered strontium ferrite (SrO-6(Fe2O3)), encased in plastic. The horizontal component of the magnetic field (perpendicular to the magnet surface) was 186±6 mT, a field strength similar to those commonly employed in therapeutic applications . Temperature changes were rigorously controlled.
EMF exposure was for 30 minutes. After 150 minutes deoxygenation of 100 µM Hb was carried out in 50 mM Hepes buffer (pH 7.2) using the reducing agent dithiothreitol (20 mM) at 22°C, and is characterized here by the passage of the spectrum from a two-peaked to one-peaked form. Figure 1 of ref [2] shows visible light absorption spectra from radio feq PEMF and static magnetic field treated hemoglobin compared with untreated hemoglobin with a peak at about 540 and 575 nm. These peaks are seen in oxy but not deoxy hemoglobin.
It should be pointed out that this 2013 study [2]
was sponsored by Ivivi Health Sciences, a company that no longer seems to be in business.
- Fig 2 Pulsed radio frequencies do nothing to increase the transition to deoxy hemoglobin.
- Fig 3 Adding the denaturing agent urea only speeds rPRF O2 release up a little bit.
- Fig 4 Release of O2 really speeds up in a static magnetic field!
Some at Ivivi Health might have been disappointed by these results. Those that are PEMF practitioners might be extremely excited by these results.
Remembering the “almost static” nature of PEMF
Radio waves are not that far from microwaves, a wavelength strongly absorbed by water as many with microwave ovens will attest. The wavelengths of ELA used in PEMF are in the 10 to 100,000 km range compared to 1 to 10 meters for radio waves!

One of the reasons the Muehsam study was so careful to control heating was because radio frequency ablation is use to heat and destroy tissue causing problems. In a sense, at `10 cycles per second, PEMF is in between a do almost nothing prf to a do something static magnetic field. [2]surely, with all the claims that PEMF increases oxygen delivery to tissue, someone has done this experiment!
Is this how PEMF works?
This post has not even attempted to explain the mathematical nature of the Mayda 2020 prediction. [1] It is very interesting to note that Muehsam and coworkers proved their prediction some three years later in regards to static magnetic fields but not rapidly changing magnetic fields associated with pulsed radio frequencies. [2] Widipedia authors have given a very limited overview of heme group containing enzymes on the heme page. Nitric oxide synthase makes vascular dilator nitric oxide. Many in the PEMF field (not discussed in this post) have attributed the oxygen delivery aspect of PEMF to nitric oxide synthase. It should be noted that NO acts by binding to the heme group guanylyl cyclase! A previous post on this site discussed the possibility of visible light interacting with guanylyl cyclase. This is a recycled image of Some cysteines in the NO and apo states of sGC.

As an added note: sGC makes cGMP, which activates protein kinase G, which activates smooth muscle myosin light chain phosphatase, with relaxes blood vessels. Heme is such an important cofactor in so many enzymes that seem to be impacted by PEMF. PRF has a small influence on O2 bound to hemoglobin. SMF has more of an influence. PEMF, somewhere in the middle, might do more. The possibility of being able to tune PEMF to a particular heme cofactor is most intriguing!
References
- Mayda S, Kandemir Z, Bulut N, Maekawa S. Magnetic mechanism for the biological functioning of hemoglobin. Sci Rep. 2020 May 22;10(1):8569. PMC free paper
- Muehsam D, Lalezari P, Lekhraj R, Abruzzo PM, Bolotta A, Marini M, Bersani F, Aicardi G, Pilla A, Casper D. Non-thermal radio frequency and static magnetic fields increase rate of hemoglobin deoxygenation in a cell-free preparation. PLoS One. 2013 Apr 12;8(4):e61752. PMC free paper
Leave a Reply