This is not a “feel good” post. Photodynamic therapy, photobiomodulation, can produce singlet oxygen. Just a little of a good thing can be healing. Over a certain threshold can lead to cell death. Three review articles have been shown to illustrate this phenomenon of why this is so. One thing that sets Red Light Therapy Home is that the company provides guidelines of when too much of a good thing is not a good thing.
What are singlet and triplet oxygen?

- Remember these atomic solar systems from school?
- Oxygen has 8 electrons to account for.
- Two have opposite spins and are found in the 1s orbital.
- Two more both with opposite spins in the 2s orbital.
- Moving up to 2p we have four electrons,
- Two of these have the same spin.
- This is how oxygen got to be a “di-radical”
A singlet state contains 0 unpaired electrons. The doublet state contains 1 unpaired electrons. The triplet state contains 2 unpaired electrons.
Note that these planetary orbitals only represent energy levels, not where one might find an electron at any given time because electrons do not orbit the nuclei like planets orbit the sun. Electrons play around in the neighborhood of the nucleus depending on their energy level and where other electrons are playing. In this discussion only energy matters rather than the places one is most likely to find an electron. Chem 3D has a very beautiful discussion of electron probability clouds. These clouds are absolutely beautiful if the ready has time to browse in awe. Murotomi 2023 Fig 1 is an energy diagram of the triplet ground state and two excited singlet states. Higher energy levels are higher up in the strata. Cui 2024 Fig 1 has a very similar energy diagram.
Many references on the Internet claim that singlet and triplet terms, that apply to many molecules, arise from old electron paramagnetic resonance experiments when quantum mechanics was much younger.
- Arises from unpaired electrons
- Due to their spin, unpaired electrons have a dipole moment

- This image was inspired by WikiReader’s YouTube video of the same name.
- The triplet state has two unpaired electrons….
- Two paired electrons in the 1st oxygen form a 1/2 π bond with the unpaired electron of the 2nd oxygen atom in the 2p “orbital”.
- Likewise, an unpaired electron from the first oxygen…
- Singlet states have no unpaired electrons.
- The bottom configuration is singlet oxygen.
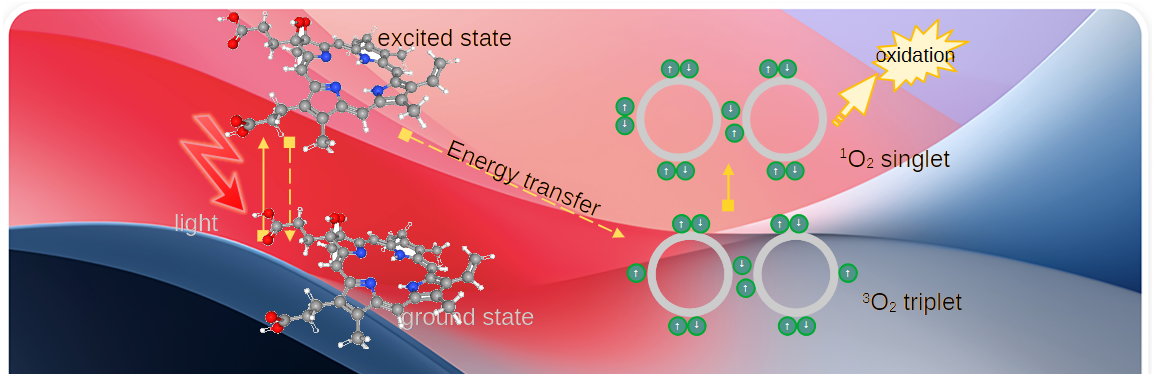
Fig 1 of Cui 2024 further illustrates this phenomenon. The ground state is like the top image with electrons in with the same spin in different orbitals forming partial π bonds. The lower energy 1O2 state has electrons with opposite spins in the same orbital and non in the other O molecule. The highest energy 1O2 is like the top state cartoon with single electrons with opposite spins.
The really cool thing about Cui 2024 Fig 1 is that the energy transitions of excitation and decay are given in nm of red and NIR light. Recall that the short wavelengths have more energy. 1270 nm is a common wavelength of fluorescence/phosphorescence of 1O2 returning to the 3O2 ground state.
Fig 2 Cui 2024 two chemical reactions of photo excited heme
Photobiomodulation depend on three elements: light, a photosensitizer, and molecular oxygen. Porphyrins, which heme is one, are widely used as photosensitizers. Like PEMF, photobiomodulation follows the same theme: small amounts of 1O2 generate oxidative stress responses that include ca2+ signaling that activate protein kinases and transcription factors. When these responses exceed the cell’s antioxidation capacity therapeutic killing , eg cancerous cells, occur. Fig 1 of Fujii 2023 says pretty much the same thing in a slightly different cartoon.
- Type 1 The electron in the excited state is transferred direly to O2 to produce superoxide O2•- or hydrogen peroxide H2O2. These two species are generally considered more reactive than singlet 1O2.
- Type 2 The energy released in the decay of the electron of the porphyrin from an excited state to the ground state changes the spin of the electrons in triplet 3O2 to singlet 1O2.
1O2 can return to the ground state in two ways: physical and chemical.
- Physical is simply heating up surrounding molecules or the release of a photon.
- Chemical is transferring energy to other molecules with bonds capable of being in the triple and singlet state just like molecular O2.
2.2. Physical Chemistry Properties of Singlet Oxygen
- The return of 1O2 to the ground state can be by the release of a 1270 nm photon. The luminescence rate constant (ke) for 1O2 is the rate at which 1O2 is consumed by the luminescence process upon return to the ground state. Fluorescence is vey short lived version of luminescence in that the return to the ground state is immediate after the source of excitation has been removed. Phosphorescence results from a longer lived excited states. Solvents, or the materials surrounding the excited molecule.
- Table 1 lists the lifetimes of porphyrin rings in different solvents, some hydrophobic, come not. The less quenching from the environment, the more likely 3O2 can react for the formation of 1O2.
- The lifetime of 1O2 is also dependent on the environment. Non radiative decay can occur by two means: (1) Groups like C-H, N-H, and O-H groups are good at absorbing kinetic collisional heating up. Reactions go faster as the temperature increases. (2) A chemical reaction with excited singlet-state β-carotene producing triplet-state β-carotene and 3O2. Other examples of chemical reactions involve actual changes in the quenching molecule such as introducing hydroxide groups into double bonds. Lipid peroxidation is one such example.
- 1O2 reacts with amines to produce free radicals and ascorbate to form hydrogen peroxide (H2O2)
- The quenching rate directly impacts the lifetime of 1O2. A fast quenching process shortens the lifetime, whereas a slow process or low quencher concentration prolongs it.
Oxidizing Activity of Singlet Oxygen
These bullet points are from the Murotomi 2023 review:
- 1O2 reacts with unsaturated fatty acids in cell membranes, Fig 5 of Fujii 2024 has causing cell death by cell membrane lipid peroxidation.
- 1O2 reacts with histidine to hydroxyimidazolone, and oxidation of tryptophan to N-formylkynurenine)
- 1O2 reacts with the thiols of amino acids methionine and cysteine. Fig 4 of Fujii 2024 has 1O2 activating cell growth by inhibiting teh phosphtase activity of protein tyrosine phosphtases (PTP) that turn off the response to growth factor receptors.
- 1O2 also induces the oxidation of guanine and causes oxidative damage of DNA .
Fig 3 of Murotomi 2023 is an illustration of different 1O2 reactions. Fig 3 of Fujii 2023 shows the oxidation of lipids, guanine base of DNA, and the amino acid cysteine.
Muronomi 2023 claim that physiological levels of 1O2 have signaling roles via reversible and irreversible thiol (in cysteine) oxidation.
Red Light Warning
Beware of any red light therapy that does not give suggestions on exposure for given conditions that include time, wavelength, and distance from the light. Too much red light can really lead to the generation of reactive oxygen species that can lead to irreversible damage to proteins. Small amounts of reactive oxygen species can lead to beneficial cell signaling. More intense red light therapy with photosensitizers is used to kill cancer cells. Much of Red Light Therapy Home’s recommendation’s are based on scientific studies. Knowing what we know now about what singlet oxygen reacts with, it is better to discuss your use of red light with your healthcare provider.
If you would like to order your own red light therapy device from Red Light Therapy Home, use this promo code for a 6% discount
Friend6-ZQDGW49R
References
- Cui S, Guo X, Wang S, Wei Z, Huang D, Zhang X, Zhu TC, Huang Z. Singlet Oxygen in Photodynamic Therapy. Pharmaceuticals (Basel). 2024 Sep 26;17(10):1274. PMC free paper
- Fujii J, Soma Y, Matsuda Y. Biological Action of Singlet Molecular Oxygen from the Standpoint of Cell Signaling, Injury and Death. Molecules. 2023 May 14;28(10):4085. PMC free paper
- Murotomi K, Umeno A, Shichiri M, Tanito M, Yoshida Y. Significance of Singlet Oxygen Molecule in Pathologies. Int J Mol Sci. 2023 Feb 1;24(3):2739. PMC free paper
Leave a Reply