This post covers two QuantumTx mouse studies that claim changes in the micobiome that includes the cecum and the pellets. What does this mean for humans wishing to change their microbiomes? The amazing thing is that the microbiome changes seen in the mice were after only 10 minute exposures of “PEMF” per week over the course of several weeks. QuantumTx has many publications demonstrating 10 minute exposures per week having dramatic metabolic improvements in mice and humans in just a few weeks. In writing this post I have come to the conclusion that the PEMF “antennas” might include not only mammalian FAD bound to cryptochrome 2 and the TRPC1 Ca2+ but perhaps some anaerobic bacterial electron transport protein(s). Potential PEMF antennas in the bacteria and the human colon are presented in this post.
The tricky formula of QuantumTx PEMF [1]
These bullet points were modified from a copy and paste from the Yap 2019 publication.
- barrages of 20 × 150 μs on and off pulses for 6 msec
- repeated at a frequency of 15 Hz.
- magnetic flux density rise (slew) maximal level within ~50 μs (~17 T/s)
- driving field amplitudes between 0.5 and 3 mT.
My calculations
- The 6 msec “barrages” contain ~35 on/off events of 170 μs, or 0.17 msec.
- With 167 intervals of 6 msec in a second, this comes out to about 5,800 on/off events per second.
- One might say about 6 kHz. This is still very low frequency, not quite radio frequency.
This is different than the radio frequency device described in the Assisi post whose device was programmed to activate Ca2+ calmodulin and all down stream pathways. There is nonetheless something “magic sauce” about this wave form that suggests a proprietary target. While some of this might be suggestive of radio frequency voltage transients coming out of dirty 50/60 Hz electricity, imagers of the wave form look much cleaner. The field amplitudes are greater too.
PEMF exposure at an amplitude of 1.5 mT was applied to C57BL/6NTac mice once per week on Sundays for 10 minutes, the usual RF frequency bursts. Treadmill exercise days were on Mondays, Wednesdays, and Fridays. [2,3]
Microbiome from which part of the colon?
The next section is going to make the case for PEMF making a mammal defecate. The QuantumTx group used a combination of mouse pellets and the mouse cecum, a homolog of our appendix, which is for them a giant fermentation chamber.

The mouse cecum is between the small intestine and colon. From my memories of my lab days is it filled with a fermenting muck that tends to be brownish green. It is basically fermenting muck, a far divergence from the well formed and often rock hard pellets further down in the colon. Wong 2025 answered my question “what microbiome?” in their protocol.
The microbiome of which part of the colon/rectum? I’ve a lab/heeler mix that makes me take her running every day. Most dog owners are used to carry bags for feces. When a dog runs, it brings home that an ecological progression occurs as fecal material moves through a quicker than humans canine GI tract.

The reader will be spared graphic images of Mindy’s bowel movements on her runs. The first movement produces a generally fully formed mass of fecal material with minimal moisture content. The second movement occurs latter during the run. It smells different, it is watery, and tends to have a different color from the first.
The journey from mouth to anus differs from mouse, to dog, to human. Anecdotally, changing the the transit time can change the attributes of what comes out of he anus in ways that extend beyond the hydration state. Giving the contents more time to exit can lead to changes in the bacterial digestion of what the mammal cannot digest and one would think the relative numbers of bacterial species.
PEMF Antenna in the Colon
Previous QuantumTx posts on this site suggest that flavin adenine dinucleotide (FAD) bound to cryptochrome 2 is the antenna for PEMF. Receipt of the signal results in the generation of reactive oxygen species via the “radical pair mechanism” These ROS activate the Ca2+ channel TRPC1. Are these two proteins colocalized in the human colon?
Colonic histology primer from Protein Atlas
The initial barrier is formed by the brush border enterocytes, mucus secreting goblet cells, and hormone secreting glandular cells (enteroendocrine cells) which respond to chemicals in the gut and secrete hormones that synapse to the enteric nervous system.
The lamina propria contains connective tissue, immune cells, and some nerve endings. The muscularis mucosa is a muscle layer just below the lamina propria. In the sub mucosa we have a layer of circular and longitudinal smooth muscle with nerve fibers running between them. Ganglia and nerve fibers between the two layer of the muscularis externa are part of the myenteric plexus (aka the Little Brain). This is part of the autonomic nervous system and control among other things; motor function, secretion, absorption, blood flow and interaction with other organs.

CRY2
A previous post on Cry2 covered a 2025 QuantumTx paper demonstrating that the circadian flavoprotein cryptochrome 2 is an antenna for the QuantumTx combination of frequency waveform and magnetic field strength. This makes he detection of Cry2 in the glandular cells and the ganglion of the myenteric plexus extremely exciting for all those that use PEMF to “get things moving.”

Cry2 also appears to be part of the muscularis mucosa. Work from the QuantumTx group suggests that the calcium channel TRPC1 is a target of reactive oxygen species produced by Cry2.
TRPC1
These images are also from Protein Atlas. We also see strong staining of glandular cells and ganglion cells of the myenteric plexus. This is again another exciting observation for those that use PEPMF for intestinal motility. It should be pointed out that the myenteric plexus controls the coordinated contraction of circular and longitudinal smooth muscle layers.

All of these imagers speak to the possibility that PEMF might do something to enhance how the gut responds to bacteria. Surely the investigators would have mentioned it if the mice evacuated the content of their colons and cecum. Perhaps the PEMF antenna is in the bacteria themselves even for the changes in the mouse physiology.
The Firmicutes/Bacteroidetes ratio`
PubMed has over 1000 peer reviewed with this search term. The F/B ratio seems to be used as a biomarker for general health.

The Wikipedia entry for Bacteriodota mentions succinic acid, acetic acid, and sometimes propionic acid as metabolic byproducts of digesting mucus, urea, and such in our GI tracts. Wikipedia authors do not have a lot to say about the metabolism of Bacillota (aka Firmicutes)
Tai 2020 10 min PEMF affects the microbiome of mice[2]
As my running dog Mindy could tell you, exercise modulates metabolism and the gut microbiome. The QuantumTx had previously shown that brief exposure to low mT PEMF plus PEMF promoted transcription of mitochondria genes via transcription factor PGC-1α transcriptional upregulation. The combination of PEMFs and exercise for 6 weeks enhanced running performance and upregulated muscular and adipose Pgc-1α transcript levels, whereas exercise alone was incapable of elevating Pgc-1α levels. The gut microbiome Firmicutes/Bacteroidetes ratio decreased with exercise and PEMF exposure, alone or in combination, which has been associated with many things in the published literature. After 2 months, brief PEMF treatment alone increased Pgc-1α and mitohormetic gene expression and after >4 months PEMF treatment alone enhanced oxidative muscle expression, fatty acid oxidation, and reduced insulin levels. See the Graphical Abstract.
| Firmicutes/Bacteroidetes ratio | -PEMF | +PEMF |
| no exercise | 1.0 | 0.8 |
| exercise | 0.8 | 0.6 |
Tai 2020 was mostly about the metabolism of the female mice:
- Figure 1 was a six month arm of the overall study in which the mice did not exercise. Fibers in the slow twitch soleus muscle were counted. They increased in number as the field strength increased from 0 mT to 2 mT. The cross sectional area of individual fibers appeared to decrease. Blood insulin decreased in mice not exercising compared to no PEMF! Smaller changes were noted.
- Figure 2 PEMF treatment accentuates the effects of weekly exercise in mice soleus and EDL slow and fast twitch fibers. Mitochondria gene master regulator Pgc-1α expression in slow twitch soleus muscle.
- Figure 3 Pgc-1α, increased in response to PEMF (irrespective of exercise) in white and brwon adipose issue. Other changes also occured.
- Figure 4 Mitohoremetic responses that include Sirt1, leptin, adiponectin, and IL-6.
- Figure 5 various measurements of microflora species diversity.
- Figure 6 LEfSe analysis revealed
| LEfSe | – PEMF | +PEMF |
| no exercise | Bacteroidetes (3 OTUs), Firmicutes (10 OTUs), Tenericutes (4 OTUs), and Proteobacteria (2 OTUs) | Tenericutes (6 OTUs) and Acinetobacteria (5 OTUs) |
| exercise | Bacteroidetes (3 OTUs), Verrucomicrobia (6 OTUs), Firmicutes (2 OTUs), and Acinetobacteria (2 OTUs). | Bacteroidetes (8 OTUs), Firmicutes (2 OTUs), Proteobacteria (4 OTUs) |
These mice showed PEMF and/or exercise enrichments of gut commensals that were liked to positive energy balance in other studies. What was not discussed was whether PEMF and/or exercise were killing off certain metabolic types of bacteria. If exercise and/or PEMF were causing the mice a diarrhea that might influence the microflora, the authors would have mentioned it as they did other experimental nuances.
Wong 2025 PEMF, fecal microbial transplants and more [3]
Can PEMF exposure not only heal metabolic issues but make better poops. The Graphical Abstract of this paper covers multiple health benefits from taking feces from PEMF exposed mice and transferring them to donor mice on high fat diets. Dried fecal pellets were collected between weeks 7 and 8 from the mice that had received weekly PEMF therapy or exercised as mentioned before. PEMF and Exercise Interventions). To account for three daily gavages and a 10% handling loss, approximately 800 mg of fecal pellets were collected per day over 10 days, yielding the required 20 mg of feces per gavage for an 8-week period. The collected fecal samples were immediately homogenized in anaerobic PBS to preserve microbial integrity and stored at −80 °C until use. Recipient mice were gavaged with the bugs thrice weekly.
Figure 1. Study design and workflow.
(a) Paradigm 1 consisted of C57BL/6 mice undertaking 8-week interventions including the following: r
- red—no treatment (−E/−P);
- blue—PEMF exposure (−E/+P; 1.5 mT for 10 min weekly);
- green—exercise (+E/−P; twice weekly); or
- black—combined treatments (+E/+P).
Fecal/cecal samples were collected from PEMF and exercise groups for subsequent fecal microbiota transplantation (FMT).
Quick overview of figures not related to this microbiome post
Figure 2 examined white adipose tissue biochemistry in PEMF and/or exercise poop donating mice as well as the white fat biochemistry in high fat diet recipient mice. Should they also be on PEMF too? Figure 3 brown adipose tissue biochemistry, Figure 4 and Figure 5 bone health, Figure 6 adipokines including leptin, Figure 7 glucose tolerance, Figure 9 and Figure 10 hepatic lipid profile.
Fig 8 good/bad Phyla ratios
Changes in the gut microbiota were evaluated using 16S rRNA gene sequencing of the fecal and cecal matter. Recall that phyla are below Kingdom and above Class in phylogenetic ranking. In both the FMT donor and FMT recipient mice, the predominant microbial phyla were Firmicutes, Deferribacteres, and Bacteroidetes. An elevated Firmicutes-to-Bacteroidetes (F/B) ratio is linked with metabolic disorders and predictive of adiposity. Clostridia are representatives of Firmicutes and Bacteroides of Bacteroidetes. A high F/B ratio is generally considered a maker of poor microbiome health. The reader is invited to consult this public access publication for exact details. Figure 8 also examined Deferribacteres/Bacteroidetes (D/B ratio) from FMT donor and FMT recipient HFD mice.
My thoughts on the QuantumTx papers
Wong 2025 claim “The Deferribacteres are pro-inflammatory bacteria that thrive in low-oxygen environments and contribute to metabolic disease.” They may also contribute to exercise induced stress. According to Wikipedia authors, Deferribacteraceae may use iron, manganese, and nitrate as terminal electron acceptors in anaerobic respiration. Wong 2025 cited literature saying that exercise stress could increase the D/B ratio. This makes sense. If the liver is breaking down trashed out red blood cells and releasing excess iron in the enterohepatic circulation. Is the D/B ratio a reason why eating too much red meat is considered bad? This site contains a post on how Arthur Pilla used radiofrequency PEMF to expedite Ca2+ with calmodulin, see the Assisi post . Could the QuantumTx RF waveform muck up the use of Fe3+ as a terminal electron acceptor by Deferribacteres? Could RF from a lap top on the lap for several hours make this happen?
Half of the battle of learning more is knowing the right search terms. Dissimilatory metal-reduction seems to be a good starting point given a good Wikipedia page on microorganisms with said function. In previous microbiome gigs, Desulovibrio sulfate reducers were of concern.
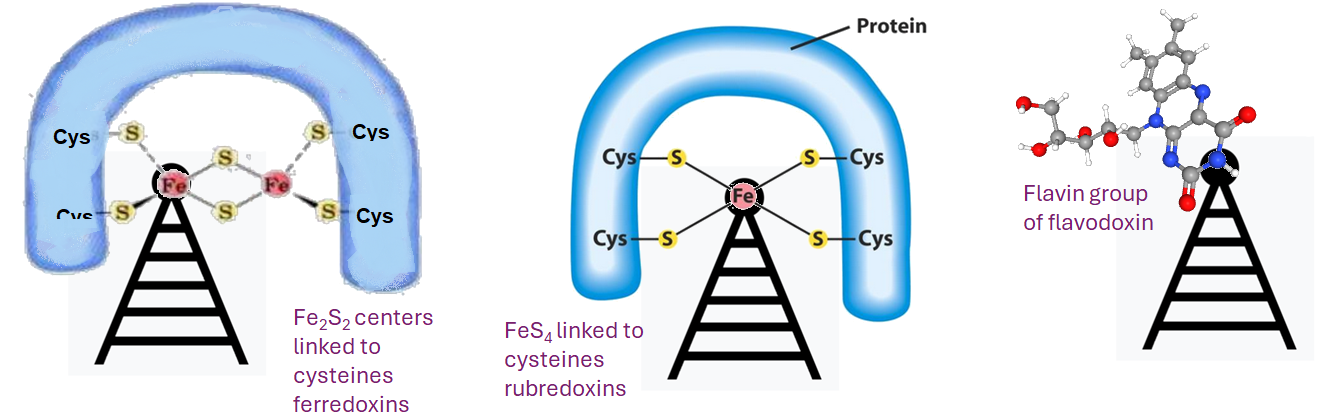
Potential PEMF antenna in a Clostridium species, Ragdale 1984
Ragsdale 1984 is an old timey paper on the isolation and characerization of redox active proteins from C. formicoaceticuum. On of my first jobs as an undergrad was washing lab ware in a lab that isolated high potential iron sulfur proteins from bacteria to characterize their redox properties spectrophotometically. They used the spectrophotometers to measure the redox/color changes. The exiting part about all of this is our search has led us to some ways in which the RF frequency wave form that QuantumTx uses might being killing off a Firmicutes phyla member Clostridia sp.
Ferredoxin, flavodoxin, and rubredoxin were purified to homogeneity from Clostridiumformicoaceticum and characterized. Variation of the iron concentration of the growth medium caused substantial changes in
the concentrations of ferredoxin and flavodoxin but not of rubredoxin. The ferredoxin has a molecular weight of 6,000 and is a four iron-four sulfur protein with eight cysteine residues. The spectrum is similar to that of other ferredoxins. The molar extinction coefficients are 22.6 x 10′ and 17.6 x 103 at 280 and 390 nm, respectively. From 100 g wet weight of cells grown with 3.6 ,uM iron and with 40 ,M iron, 5 and 20 mg offerredoxin were isolated, respectively. The molecular weight of rubredoxin is 5,800 and it contains one iron and four cysteines. The UV-visible absorption spectrum is dissimilar to those of other rubredoxins in that the 373 nm absorption peak is quite symmetric, lacking the characteristic 350-nm shoulder found in other rubredoxins. The flavodoxih is a 14,500-molecular-weight protein which contains 1 mol offlavin mononucleotide per mol of protein. It forms a stable, blue semiquinone upon light irradiation in the presence of EDTA
or during enzymatic reduction. When cells were grown in low-iron medium, flavodoxin constituted at least 2% of the soluble cell protein; however, it was not detected in extracts of cells grown in high-iron medium. The rubredoxin and ferredoxin expressed during growth in low-iron and high-iron media are identical.
- Ferredoxins can be reduced by using NADH (-320 mV) or H
2 (-414 mV), but these processes are coupled to the consumption of the membrane potential to power the “boosting” of electrons to the higher energy state. This Wikipedia is backed up with an excellent peer reviewed publication. [4] - Rubredoxins are not high potential F-S proteins per se. They contain four Cysteine residues that form disulfide bonds with FeS4. Some of the not so well referenced comments of Wikipedia author(s) give a one electron reduction redox potential range of +50 mV to -50 mV. A comment was also made that the d-shell electron of the Fe remaining “high spin” regardless of the redox status.
- Flavodoxins are small, soluble electron transport proteins that contain Flavin mononucleotide as a cofactor. FAD can exist in this enzyme as a quinone, hydroquinone, and semiquinone. What this means in the radical pair mechanism that QuantumTx as part of their mammalian formula is unknown. It is noteworthy that Wikipedia authors mention Helicobacter pylori as a bacterium that uses flavodoxin.
References
- Yap JLY, Tai YK, Fröhlich J, Fong CHH, Yin JN, Foo ZL, Ramanan S, Beyer C, Toh SJ, Casarosa M, Bharathy N, Kala MP, Egli M, Taneja R, Lee CN, Franco-Obregón A. Ambient and supplemental magnetic fields promote myogenesis via a TRPC1-mitochondrial axis: evidence of a magnetic mitohormetic mechanism. FASEB J. 2019 Nov;33(11):12853-12872 PMC free paper
- Tai YK, Ng C, Purnamawati K, Yap JLY, Yin JN, Wong C, Patel BK, Soong PL, Pelczar P, Fröhlich J, Beyer C, Fong CHH, Ramanan S, Casarosa M, Cerrato CP, Foo ZL, Pannir Selvan RM, Grishina E, Degirmenci U, Toh SJ, Richards PJ, Mirsaidi A, Wuertz-Kozak K, Chong SY, Ferguson SJ, Aguzzi A, Monici M, Sun L, Drum CL, Wang JW, Franco-Obregón A. Magnetic fields modulate metabolism and gut microbiome in correlation with Pgc-1α expression: Follow-up to an in vitro magnetic mitohormetic study. FASEB J. 2020 Aug;34(8):11143-11167. free paper
- Wong, J.K.C.; Patel, B.K.; Tai, Y.K.; Tan, T.Z.; Khine, W.W.T.; Chen, W.C.; Kukumberg, M.; Ching, J.; Lee, L.S.; Chua, K.V.; et al. Fecal Microbiota Transplantation from Mice Receiving Magnetic Mitohormesis Treatment Reverses High-Fat Diet-Induced Metabolic and Osteogenic Dysfunction. Int. J. Mol. Sci. 2025, 26, 5450. free paper
- Ragsdale SW, Ljungdahl LG. Characterization of ferredoxin, flavodoxin, and rubredoxin from Clostridium formicoaceticum grown in media with high and low iron contents. J Bacteriol. 1984 Jan;157(1):1-6. PMC free paper
- Buckel W, Thauer RK. Flavin-Based Electron Bifurcation, Ferredoxin, Flavodoxin, and Anaerobic Respiration With Protons (Ech) or NAD+ (Rnf) as Electron Acceptors: A Historical Review. Front Microbiol. 2018 Mar 14;9:401. PMC free paper
Flavoredoxin and ferroredoxin have been reviewed by Buckel and Thauer 2018 [5]. Some many potential PEMF antenna in our colons. Mitochondria are said to have evolved from bacteria all packed with proteins with functional groups with unpaired electrons that can potentially respond to RF PEMF. That QuantumTx PEMF can change microbiomes is truly incredible. This post has made a case that RF PEMF in our colon can be antenna. How much more so could the microflora themselves? Can our GI tract packed with potential antenna respond to less intense RF PEMF from dirty electricity?
Leave a Reply