Part of the long journey of learning how PEMF interacts with molecular oxygen, O2, is discarding the notions of electrons, atoms, and molecules we learned in grade school.
New Terms
- Standing wave as defined by Wikipedia authors is a sinusoidal occurrence whose peak amplitude oscillates in time but not in space. One example is stationary waves of water in a vibrating container. Another example is a vibrating string of a violin. Standing waves occur when waves from one direction combine with waves moving in space in the other direction. One can observe constructive and destructive interference.
- Valence bond theory, molecular bonds are the overlap of outer (valence) atomic orbitals as we learned in grade school.
- Molecular Orbital theory states that electrons reside in orbitals involving the entire molecule!
- ψ is the standing wave function with a positive (think up) and negative (think down) function. ψ2 is the probability of finding an electron in a given location. Robert Belford University of Arkansas at Little Rock. Link.
- Quantum numbers of an electron in an atom of hydrogen include: principal (shell or orbital), azimuthal (angular momentum along an axis in space), Magnetic (from angular momentum), and spin.
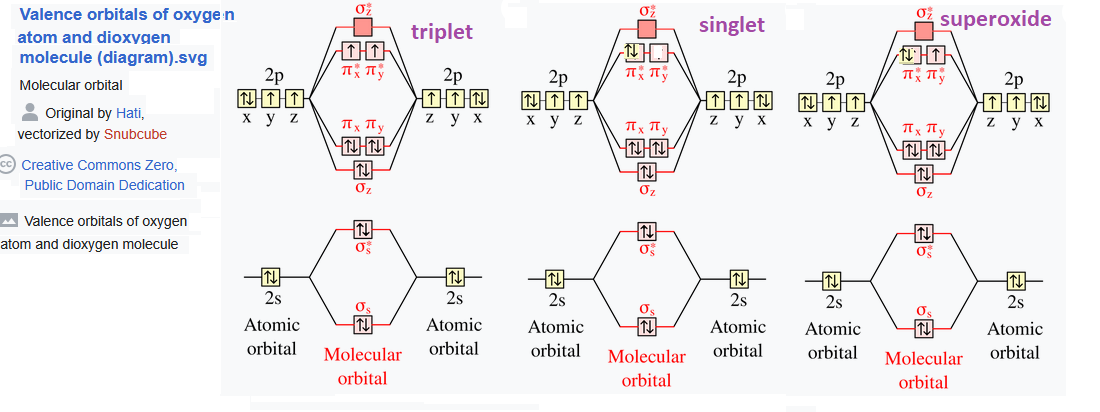
O2 atomic orbitals from K-12 school

While atomic orbitals are not exactly like planetary orbitals, the element of spin and angular momentum are similar. In addition, there’s a magnet component too from a charged something in motion. We get into wavelike properties.
Two unpaired electrons are considered to be in the triplet state, two unpaired electrons doublet, and zero unpaired electrons the reactive singlet state. Triplet O2 is paramagnetic.

This image was adapted from a WikiReader YouTube video on Triplet Oxygen. These so called 1/2 π are anti-bonding in character. The Lewis Dot structure from which this grade school atomic orbital molecule was adapted from occurs around 3:55 minutes into the video.
Hybrid orbitals
LibreTexts was a source of this imagry as well as Wikimedia Commons as indicated.

Robert Bedford in his Libretexts on Molecular Orbital Theory indicated that the z-axis is often taken as the axis between the two nuclei. As we will learn now, things are way more complicated and exciting.
ChemTube3D
The triplet O2 ChemTub3D site is interactive and allows the user to rotate the triplet oxygen molecule to get a better view of the 2πx subshell of electrons. The 2πy is almost the same in terms of energy and shape.
Triplet O2 molecular orbital

This image on ChemTube3D molecular O2 energy levels. Libretexts on Molecular Orbital Theory had an interesting take on +/- up/down spins of the electrons in color code. The main “take home” lesson is that anti-bonding configurations have a higher energy level. In singlet O2 the half filled anti-bonding same spin electrons are spin paired only at a higher energy level.
2pxπ
We kind of created the p shell sigma bond along the z-subshell. Only the x and y are available for π bonds

Note that the shape of the 2πx is vaguely peanut rather than a 3D ellipse. According to Nick Greeves, the site creator, Nick Greeves, singlet O2 occupies the same space only with spin paired electrons.
2pyπ
We still see the vaguely peanut shape of the π bonds. The orientation is different from those in the px subshell.

So what does this have to do with PEMF or even magnetic field?
Weak magnetic fields flavoproteins, O2 and reactive oxygen species
Austvold CK, Keable SM, Procopio M, Usselman RJ. Quantitative measurements of reactive oxygen species partitioning in electron transfer flavoenzyme magnetic field sensing. Front Physiol. 2024 Feb 2;15:1348395. PMC free paper
Up to this point this post has been dispelling the notion of “solar system” atomic and molecular orbitals. This particular publication examined the mitochondrial flavin adenine dinucleotide component of the electron transport chain. At some points FAD may have an unpaired electron during its reduction and oxidation duty cycle. These authors used static magnetic fields of 20 nT and 50 μT. The stronger field favored H2O2 production and the weaker field superoxide O2 •− triplet products relative to 50 µT. “The method presented here illustrates the potential of a novel magnetic flavoprotein biological sensor that is directly linked to mitochondria bioenergetics and can be used as a target to study cell physiology.”
E-FlredH2(↑↓) + O2 (↑↑)➔ E-FlredH–(↑↓) + O2 (↑↑) ➔ 3[E-FlsqH●(↑)…(↑)πx (↑↓) πyO2●-] ⇔ 1[E-FlsqH*(↑)…(↓)πx (↑↓) πxO2●-]
The ” ● ” are radical electrons. E is an enzyme containing a flavin (Fl) group. There is some H+ transfer going on since this is a mitochondrial flavo protein. Note the equilibrium between the two complexes (gray highlight) containing superoxide, O2●- Also note some spin flipping between (↑)πx and (↓)πx. This makes the 3D models of O2 orbitals on ChemTube3D all the more intriguing.
Tying things up
This is only the start of a very long journey. This post aimed to dispel the notion that electrons are like planets orbiting a sun. While there might be a spin and angular momentum, these factors bring in a magnetic function. Diving in deeper, there are three dimensional shapes of where one is likely to find the standing wave essence of an electron. Like wave in a vibrating glass of water, there are constructive (bonding) and destructive (anti-bonding) regions. Triplet O2 is paramagnetic meaning that it can respond to a magnetic field. PEMF and static magnet practitioners like to say that their tools increase the biological processes involving oxygen. If one really has an imagination, these electron probability clouds resemble the shapes of chambers in a gothic cathedral. Viewing the complexities of electrons of oxygen, it is easy to feel like a medieval peasant wandering into a gothic cathedral. Health regulatory agencies rightly frown on making unsubstantiated claims regarding static magnetic fields and PEMF. What we know certainly advocates for a relentless search for more knowledge!
Leave a Reply