The QuantumTx group demonstrated that PEMF inhibits MCF7 cancer cells and spares MCF10a normal cells. The main difference is that MC7 cells have chromosome compartmentalization that favors Wnt signaling. Wnt signaling is thought to be a mechanism of PEMF bone healing. What is Wnt signaling? The PEMF gated Ca2+ might be involved. We know that TRPV1 opening upregulates its own transcription. What else is in the TRPC1 gene neighborhood?
Barutcu 2015 Wnt pathway in MCF7 vs MCF10 breast cancer cell lines
Background
Ainoa Planas Riverola has a good piece on 3D DNA structure and regulation.
- Chromosomes reside in specific regions of the nucleus, much like books on a bookshelf.
- Compartments. Inside a chromosome many like to call A and B compartments which have open compartments of genes being transcribed. This spatial segregation of genes into different compartments provides an additional layer of control over gene regulation, ensuring that genes with similar functions or expression patterns are co-regulated and functionally coordinated. Human Wnt5A is the image for this section.
- TADs, TADs, topically associated domains, are neighborhoods within the DNA that contain genes with non-protein coding regulatory elements that physically interact with each other.
- Loops may contain promoters just upstream of the protein coding part of the gene and the very much upstream enhancer.
Higher-order chromatin structure is often perturbed in cancer and other pathological states. Although several genetic and epigenetic differences have been charted between normal and breast cancer tissues, changes in higher-order chromatin organization during tumorigenesis have not been fully explored. To probe the differences in higher-order chromatin structure between mammary epithelial and breast cancer cells, we performed Hi-C analysis on MCF-10A mammary epithelial and MCF-7 breast cancer cell lines.
Results
Our studies reveal that the small, gene-rich chromosomes chr16 through chr22 in the MCF-7 breast cancer genome display decreased interaction frequency with each other compared to the inter-chromosomal interaction frequency in the MCF-10A epithelial cells. Interestingly, this finding is associated with a higher occurrence of open compartments on chr16–22 in MCF-7 cells. Pathway analysis of the MCF-7 up-regulated genes located in altered compartment regions on chr16–22 reveals pathways related to repression of WNT signaling. There are also differences in intra-chromosomal interactions between the cell lines; telomeric and sub-telomeric regions in the MCF-10A cells display more frequent interactions than are observed in the MCF-7 cells.
Conclusions
We show evidence of an intricate relationship between chromosomal organization and gene expression between epithelial and breast cancer cells. Importantly, this work provides a genome-wide view of higher-order chromatin dynamics and a resource for studying higher-order chromatin interactions in two cell lines commonly used to study the progression of breast cancer. ”
A critical take, looking at the Wnt5A gene neighborhood
These things are fun to play with. Let’s take the human Wnt5A gene that will enter later in this post. One can take any gene at random and find related genes in the so called compartment. It was not clear from the Rivola page just how big one of these comparments in.

The bottom line is that there is nothing simple about cancer. What separates the MCF7 breast cancer cells from normal MCF10 breast epithelial cells seems to be different from simple gene mutations.
Martinez-Marin 2025 the Wnt/Frizzled pathways
Please consult the Wnt pathway link to learn about just how complicated the three Wnt frizzled pathways really are.
The three Wnt pathways
- The canonical pathway is one of those examples in biology in which a transcirpition factor, in this case β-catenin, is under a constant state of synthesis and degradation, until an extracellular signal tells the degradation to stop so as to enable new gene transcription. Lrp5/6 is the coreceptor for Wnt with FZD.
- The second pathway is the Planar Cell Polarity, or PCP. ROR/Ryk is the co-receptor for Wnt isoforms. The end game is to activate JNK and ROCK kinases leading to the phosphorylation of the transcription factor c-June. To make matters more confusing, Rac1 and RhoA, small GTPase proteins are part of the signaling. Rac and Rho were famous for regulation of the cytoskeleton in cell migration in the early days.
- In the Ca2+ non canonical pathway ROR.Ryk coupled to Fzd are the Wnt receptor. Wnt binding activates heterotrimeric G-proteins,: → phospholipase C (PLC) .→IP3 + and diacylglycerol (DAG) .→ Ca2+ stores,.→ protein kinase C (PKC) .→ Ca2+/calmodulin-dependent protein kinase II (CaMKII), calcineurin .→ NFAT. In the Martiniwez-Mari cartoon Wnt pathway l pay very close attention to the non specified Ca2+ channel on the cell membrane that is presumably regulated by on of the protein kinases, PKC and/or CamKII. In the QuantumTx model, this could be TRPC1.
Human isoforms of Wnt and Frizzled

The Hu 2024 review has covered the consequence of mutations and total loss o Wnt isoforms in bone function.
General thoughts on G-protein coupled receptors
On a personal note, a few years ago I was studying the cannabinoid 1 and 2 members of the G protein coupled receptor superfamily. Partial agonists, ligands that activate a receptor only partially as much as the main agonist, came up. CBD is well known in popular culture for only partially activating the CB1 receptor. An inverse agonist binds to a receptor and causes an opposite change to the normal agonist. CB1 and CB2 form heterodimers with all sorts of other G protein coupled receptors with different signaling outcomes.
Ma and Wang 2006, the complicated role of cGMP
This study used mouse F9 teratocarcinoma cells . click for summary figure.
- Wnt5a stimulated a decline in intracellular cyclic GMP and thus attenuated PKG activity. Note Gσt2 in the cartoon. This seems to stand for transducin, part of the G protein signaling in the retina that activates phosphodiesterase, that breaks down cGMP.
- Robust Ca2+ mobilization was enabled. The increase in Ca2+ mobilization made possible by a decline in intracellular cyclic GMP and a corresponding reduction in the activity of PKG.
- Elevation of intracellular cyclic GMP either by inhibition of the cyclic GMP phosphodiesterase or by addition of cell-permeable cyclic GMP analogue, 8-Br-cyclic GMP, effectively blocks the Ca2+ mobilization in response to Wnt5a. The bulk of Ca2+ mobilized, however, appears to require signaling from Wnt via cyclic GMP PDE/cyclic GMP/PKG (Fig. 5).
A rather lengthy and well referenced discussion of intracellular Ca2+ buffering is available in the public access publication of Wa and Wang 2006.

PEMF and the Wnt pathway
Most of the 23 peer reviewed publications on Wnt and PEMF on PubMed concern the skeletal system. Wnt controls the epithelial–mesenchymal transition (EMT) A table from xue2024 showing some of the Wnt related transcripts.
Crocetti 2013, using PEMF to disable HCF7 cells
This is one of the QuantumTx foundation papers in which they seem to have established part of the “secret sauce” formula.
PEMF Exposure System
The ELF-PEMF (extremely low frequency – pulsed electromagnetic field) bounds were within 5kHz and 5 mT. The goal was to maximize exposure volume while minimizing mechanical vibrations. The exposure chamber consisted of a structural framework within a ventilated µ-metal housing serving to shield against external magnetic fields while minimizing sound and such. Field non-uniformity does not exceed 4 % within the sample exposure volume. This study used magnetic fields of 2.0, 3.0 and 5.0 mT. Magnetic field uniformity was taken into consideration in the protocol development.

Panels C and D from supplemental figure 1. Several waveforms were examined for peak acceleration within the 6 msec bursts.
The results in bullet point summary
- Supplemental Fig 1 describes a lot of the optimization of the Quantum Tx group. There are some nice images of magnetic fields within the cell culture chamber not presented in this post.
- sFig2 3mT is better than 1 and 5 mT. Panel 1C in particular gives a very nice view of the 5 msec kHz pulses. Different waveforms were also examined. Non uniformity w u = |B – B0| / B0 , was a consideration in method development.
- Figure 1, 60 min exposure at 2, 3, and 5 mT pulses results in more dead MCF7 cells than the normal MCF10 cells. Dead was measured by diffusion of trypan blue past rips in the cell membrane.
- Figure 2 Sometimes the goal is not to kill cancer cells but to simply keep them from dividing. 60 and 90 min 3mT PEMF killed some but also slowed the total cell counts over the course of three days.
- Figure 3 takes another look at MCF7 cell killing that became particularly pronounced by day 3.
- Figure 4 looked at DNA strand breaks at 2.3. and 5 mT and 60 and 90min exposures with the same results.
- Figure 5, same exposures. This time cell killing was determined by annexin V staining for apoptosis, programmed cell death. Same results
- Figure 6 During apoptosis, cell membranes can bleb off. This can be detected by electrical impedance known as Impedance flow cytometry (IFC). The same PEMF exposures were used with the same results.
- Figure 7 Impedance flow cytometry can also be used to assess metabolic status. MCF10 normal breast cells were apparently benefitted by PEMFs as determined by IFC analysis at 9 MHz. In this case the impedance, complex resistance, is to a sinusoidal current of 9 MHz.
- Figure 8, the cells were made sick with hydrogen peroxide to confirm the ability of IFC to detect metabolically incompetent cells.
- Figure 9 Another flow cytometry assay using annexin V as a marker for apoptosis. The MCF10 controls entered in.
Sukumar 2024, synergy with the chemotherapy agent doxorubicin
TRPC1 expression in human breast cancers was found to be positively correlated with the proliferative capacity. TRPC1 expression correlated with the ability of brief magnetic exposure to enhance the uptake of the chemotherapeutic agent, doxorubicin (DOX), into cancer cells. Muscle tissue is also a target of dox’s ability to intercalate into double stranded DNA. With PEMF, the concentration of dox for 50% killing was less in breast cancer cells than in cultured muscle. The mechanism of TRPC1 enhancing dox entry was not determined.
Jiang 2019 Shao 2021, where the Wnt classical pathway comes in
The canonical Wnt (Wnt/β-catenin) signaling pathway is known to be crucial in regulating bone formation and development. Bone loss can occur after spinal cord injury. PEMF is used for bone healing. The PEMF waveform used in this experiment consisted of
- a 5 msec pulsed burst
- pulse width, 0.2 ms pulse wait, 0.02 ms; have 22.7 of these mini-pulses in 5 msec bursts… The frequency within the bursts is about 4.5 kHz.
- burst wait, 60 ms; pulse rise, 0.3 ms; pulsefall, 2.0 ms)
- at a frequency of 15 Hz.
- peak intensity of magnetic field about 2 mT.
After spinal cord injury, rats in the treatment group were exposed to two hours of PEMF per day for eight weeks. Real time PCR (quantitative) was used to measure levels of gene transcripts in the tibiae in response to spinal cord injury, PEMF, and the combination of the two.
- Wnt1 (P<0.001, -60.2%), SCI ↓ compared to control
- Wnt3a P<0.01, -37.7%)↓
- Wnt10b ( P<0.001, -60.2%) ↓
- p-GSK-3β/GSK-3β protein (P<0.001, -62.4%) ↓
- β-catenin protein( P<0.001, -67.7%). ↓
- Wnt1 (P<0.001, +140.3%) ↑ compared to spinal cord injury
- Wnt10b (P<0.001, +208.1%) ↑
- Wnt3a (P>0.05, +17.1%). no significant change
- p-GSK-3β/GSK-3β protein (P<0.001, +137.5%) ↑
- β-catenin protein (P<0.001, +178.5).↑
Jiang 2018 used a Glucocorticoid-Induced osteoporosis Rat model. Rats were treated wtih calcium and/or PEMF. In this case the PEMF was 50Hz at 4 mT for 40 minutes per day for 12 weeks.
- Fig 1, large increase in OPG/RANKL mRNA and protein even over the control
- Fig 2, large but variable increase in Wnt10a mRNA and a decrease in fatty acid binding protein mRNA.
- Fig 3, Many statistically significant changes in protein levels were seen. A decrease by about half in the protein levels of C/EBPα, FABP4 could be argued to be significant in terms of magnitude as well.
“BMSCs can differentiate into osteoblasts, adipocytes, myocytes, and chondrocytes [34]. Excessive use of GCs can disturb lipid metabolism homeostasis directly by GCs inducing BMSCs differentiation into adipocytes. GCs also can upregulate the expression of PPAR-γ, downregulate the Runx2 to break the dynamic balance between adipogenesis and osteogenesis of BMSCs, and lead to fat tissue accumulation in bone marrow; eventually the degree of osteoporosis was increased [7, 15]. PPAR-γ is an adipogenic gene, and it plays a pivotal role in the regulation of adipogenesis and lipid metabolism homeostasis in synergy with another key adipogenic transcription factor C/EBPα. PPAR-γ and C/EBPα positively activate the transcription of each other [17]. FABP4 is a marker of mature adipocyte. On the contrary, Runx2 is an osteogenic gene, and it is a master regulator of BMSCs differentiation into osteoblast and RANKL expression in osteoblasts [35]. Furthermore, Runx2 is a transcriptional target of canonical Wnt signaling pathway and it involves negative regulation of PPAR-γ expression

In the canonical Wnt pathway,
- Wnts bind to specific frizzled receptors and LRP5/6
- This inactivates glycogen synthase kinase 3β (GSK-3β) so that β-catenin cannot be tagged for degradation by phosphorylation.
- Spinal cord injury downregulated the expression of canonical Wnt ligands (Wnt1, Wnt3a and Wnt10b)
- SSCI also decreased p-GSK-3β and β-catenin expression.
- PEMF for 8 weeks reversed this down regulation. .
Hao 2024 the TRPC1/Wnt connection
- These authors produced a transgenic mouse line expressing porcine TRPC1. Tg-pTRPC1 increased mouse muscle mass and endurance, and accelerated muscle regeneration.
- The muscle transcription factor Prrx1 promoted the expression of TRPC1, NFATC2 and NFATC2IP. CP is for “cytoplasmic.” IP is for “interacting protein.” According to UniProt, human Prrx1 has three unique phosphorylation sites and one acetylated lysine.
- TRPC1 regulated muscle growth via activating Wnt/β-catenin and Wnt/Ca2+ pathways.
Wnt and PEMF: concluding thoughts
- Preliminary work from the QuantumTx group (Crocetti 2013) demonstrated PEMF induced apoposis in MCF7 cells while sparing MCF10 cells. Sukumar 2025 demonstrated TRPC1 expression in MCF7 cells and in patient breast cancer cells and that TRPC1 expression synergized with PEMF to increase the uptake of doxorubicin.
- Barutcu 2015 demonstrated changes in the chromosome compartments favoring Wnt signaling genes in the MCF7 cells. The Martinez-Marin review described the three versions of Wnt signaling. The non-canonical Ca2+ has components in common with mito hormetic signaling.
- Looping back to the Wuantum Tx work with MCF7 Wnt open compartment cells, Hung 2014 demonstrated a role of Wnt5A in the doxorubicin efflux pump ABCB1.
- Wnt signaling is very much involved with bone health and growth. Hung 2014 and Jiang 2016 found that their versions of PEMF increased transcription of Wnt pathway proteins.
- Hao 2024 demonstrated a link between Ca2+ channel TRPC1 and Wnt/β-catenin and Wnt/Ca2+ pathways. Wang 2006 provided evidence for the need to inhibit the cGMP dependent protein kinase PKG. This is where the PEMF activation of TRPC1therapy might come in: overcoming activation of phosphodiesterase that leads to the inactivation of PKG. The Assisi loop, covered on this site, is engineered for facilitating the binding of Ca2+ to calmodulin with activates nitric oxide synthase, which activates guanylyl cyclase, which activates PKG.
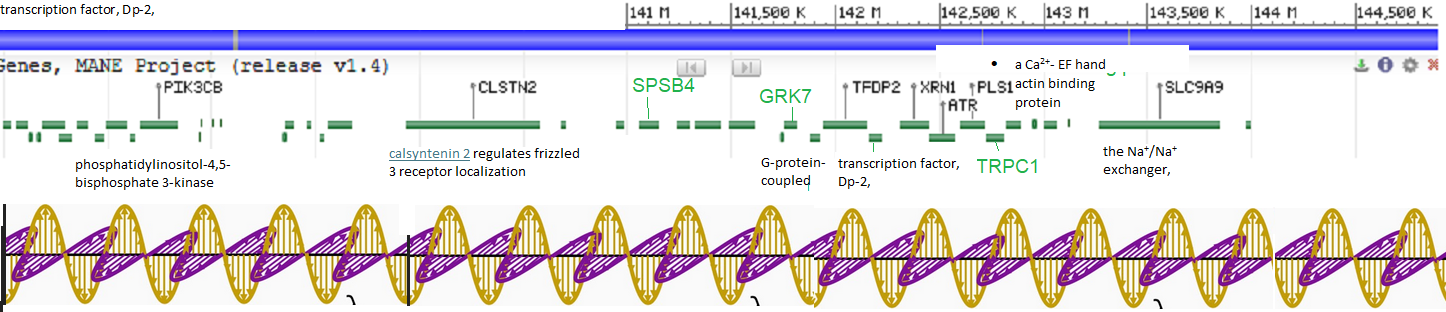
Another possibility entirely is that the feed forward expression of TRPC1 opens up the chromosome 3 compartment for other proteins of interest, in the featured image, which is of the human chromosome 3 locality of TRPC1. The mouse version is not that much different.
References
- Barutcu AR, Lajoie BR, McCord RP, Tye CE, Hong D, Messier TL, Browne G, van Wijnen AJ, Lian JB, Stein JL, Dekker J, Imbalzano AN, Stein GS. Chromatin interaction analysis reveals changes in small chromosome and telomere clustering between epithelial and breast cancer cells. Genome Biol. 2015 Sep 28;16:214. PMC free paper
- Crocetti S, Beyer C, Schade G, Egli M, Fröhlich J, Franco-Obregón A. Low intensity and frequency pulsed electromagnetic fields selectively impair breast cancer cell viability. PLoS One. 2013 Sep 11;8(9):e72944. PMC free paper
- Hao X, Fu Y, Li S, Nie J, Zhang B, Zhang H. Porcine transient receptor potential channel 1 (TRPC1) regulates muscle growth via the Wnt/β-catenin and Wnt/Ca2+ pathways. Int J Biol Macromol. 2024 Apr;265(Pt 1):130855.
- Hu L, Chen W, Qian A, Li YP. Wnt/β-catenin signaling components and mechanisms in bone formation, homeostasis, and disease. Bone Res. 2024 Jul 10;12(1):39. PMC free paper
- Hung TH, Hsu SC, Cheng CY, Choo KB, Tseng CP, Chen TC, Lan YW, Huang TT, Lai HC, Chen CM, Chong KY. Wnt5A regulates ABCB1 expression in multidrug-resistant cancer cells through activation of the non-canonical PKA/β-catenin pathway. Oncotarget. 2014 Dec 15;5(23):12273-90. doi: 10.18632/oncotarget.2631. free paper
- Jiang Y, Gou H, Wang S, Zhu J, Tian S, Yu L. Effect of Pulsed Electromagnetic Field on Bone Formation and Lipid Metabolism of Glucocorticoid-Induced Osteoporosis Rats through Canonical Wnt Signaling Pathway. Evid Based Complement Alternat Med. 2016;2016:4927035. PMC free paper
- Ma L, Wang HY. Suppression of cyclic GMP-dependent protein kinase is essential to the Wnt/cGMP/Ca2+ pathway. J Biol Chem. 2006 Oct 13;281(41):30990-1001. free paper
- Martinez-Marin D, Stroman GC, Fulton CJ, Pruitt K. Frizzled receptors: gatekeepers of Wnt signaling in development and disease. Front Cell Dev Biol. 2025 May 1;13:1599355. PMC free paper
- Shao X, Yan Z, Wang D, Yang Y, Ding Y, Luo E, Jing D, Cai J. Pulsed Electromagnetic Fields Ameliorate Skeletal Deterioration in Bone Mass, Microarchitecture, and Strength by Enhancing Canonical Wnt Signaling-Mediated Bone Formation in Rats with Spinal Cord Injury. J Neurotrauma. 2021 Mar 15;38(6):765-776. Sci-Hub free paper
- Sukumar VK, Tai YK, Chan CW, Iversen JN, Wu KY, Fong CHH, Lim JSJ, Franco-Obregón A. Brief Magnetic Field Exposure Stimulates Doxorubicin Uptake into Breast Cancer Cells in Association with TRPC1 Expression: A Precision Oncology Methodology to Enhance Chemotherapeutic Outcome. Cancers (Basel). 2024 Nov 18;16(22):3860. PMC free paper
Leave a Reply