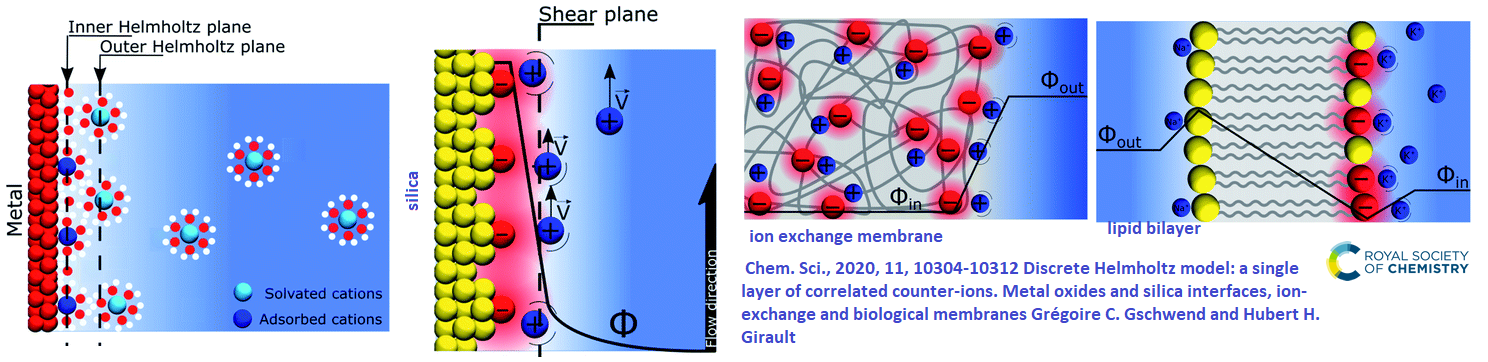
Professional organizations like MagnaWave and AOPP claim that PEMF relieves inflammation. How this happens is largely unknown. The featured image came from a review on how charges in solution line up on surface charges. from metals, to metal oxides, to ion exchange membranes, and finally biological membranes. Some images of skin and bone joints are shown. Are these like ion exchange membranes that benefit from stirring up to aid in detoxification? What about counter ions around our phospholipid cell membranes? What are the other hypotheses concerning how PEMF reduces inflammation?
An excellent PEMS in the journal of Vet Science [1]
No where does this review mention inflammation specifically, but is does give many potential mechanisms of action
Faraday’s law of induction ε=dNΦB/dt
Faraday’s equation describes how a changing magnetic field dNΦB that changes with time dt can create an electromotive force that can cause charges to move in a wire or in a tissue. Perhaps inflammation is reduced by clearing stuff out of tight spaces

This brings us back to the streaming potential in bone growth that could also be inflamed tissue.
Ca2+ channels and nitric oxide
This review covered the role of Ca2+ influx without specifying how, Ca2+ ➔binds to calmodulin➔Ca2+Cam binds to and activates nitric oxide synthase➔nitric oxide synthase makes NO➔NO binds to guanylyl cyclase➔guanylyl cyclase makes cGMP➔cGMP activates protein kinase G➔PKG activates numerous cell signaling pathways. One of the references, Rohde 2015 , came to the conclusion that “pulsed electromagnetic field therapy significantly reduced postoperative pain, inflammation, and narcotic use following TRAM flap breast reconstruction, paralleling its effect in breast reduction patients. Both studies also report a significant reduction of interleukin-1β in the wound exudate, supporting a mechanism involving a pulsed electromagnetic field effect on nitric oxide/cyclic guanosine monophosphate signaling, which modulates the body’s anti-inflammatory pathways. Adjunctive pulsed electromagnetic field therapy could impact the speed and quality of wound repair in many surgical procedures.”
Gene expression
PEMF may increase the expression of heat shock protein 70 and adenosine receptors. This particular review did not go into the details of HSP70 and adenosines in the process of inflammation. [1]
Arthritis, adenosine receptors, and 75 Hz 1.5 mT PEMF [2]
This cell culture study treated T/C-28a2 chondrocytes or hFOB 1.19 osteoblasts with ± IL-1β at 1 ng/ml for 48 hours, in the absence or in the presence of PEMFs for the last 24 hours. The 75 Hz PEMF 1.3 ms pulses had magnetic fields of 1.5±0.1 mT for chondrocytes and 2.5±0.2 mT for osteoblasts. These treatments double mRNA and protein levels of the adenosine A2A and A3A receptors for both cell types. A1A and A2B receptor expression was unaffected. The increase in A2A and A3A adenosine receptors were revealed by binding studies to be functional. Cotreatment of these two cell types with PEMF and IL-1β synergistically increased adenosine binding to the cells. Naturally there is a down stream effect on adenylyl cyclase, the maker of the second messenger cAMP. Control of AC is like driving with one foot on the brake and the other on the accelerator. The blue spur compound forskolin is sometimes used to turn on AC in absence of receptor signaling.
As expected, the A2AAR agonist CGS 21680 (100 nM) elicited an increase of cAMP as would be suggested in the featured image. A2AAR agonist CGS 21680 enhanced proliferation of both chondrocytes and osteoblasts in a manner that was potentiated by PEMF exposure. An A3A agonist was without effect on proliferation. In both the cell line examined, CGS 21680 and Cl-IB-MECA were able to significantly decrease the IL-1β-stimulated production of the inflammatory mediators IL-6, IL-8 and PGE2. The capability of both A2A and A3AR agonists to mediate anti-inflammatory effect suggests the involvement of different downstream signaling pathways. This is because A2A turns on AC while A3A turns it off. The authors reminded us that A3A agonsists also increase PI3 kinase that was discussed in a previous PEMF post on eNOS and PI3 kinase. PEMF exposure to potentiate the anti-inflammatory effects mediated by A2A and A3AR agonists.
There may be other moving parts in this PEMF story. A2A and A3A receptor agonists had no effect on osteoprotegerin production, PEMF significantly increased this exposure resulted in a significant increased release of this bone protective factor
QMR is a non-ionizing, low-potency technology that uses high-frequency waves in the range between 4 and 64 MHz delivered through alternating electric fields. QMR develops quanta of energy capable of breaking molecular bonds without increasing the kinetic energy of the affected molecules.
A summary of rheumatoid arthritis studies [3]
These authors favor ions on the surface of the cell membrane as being the antennae for the PEMF magnetic field. Indeed, free electrons in the molecular orbitals of wires are the antenna for radio frequency electromagnetic radiation. The nice thing about this review is that it summarizes papers that have used similar frequencies to those available on the MagnaWave Semi-10
| Authors | Frequency (Hz) | Field strength (mT) | Time of exposure | Outcome |
|---|---|---|---|---|
| Chen et al. | 15 | 2 | 8 h/day | Increased cartilaginous matrix deposition and other bone markers. Sox9 transcription factor |
| Linnemann et al. | 16, walking is 15 Hz | 6–282 µT | 7min | Neutrophil extracellular trap markers due to PMA, H2O2 ⇓ |
| Vinhas et al | 5-17 | 1.5,4,5 | 10-50% 1 hr | human tendon cells IL-1β inflammatory cytokines ⇓, anti-inflammatory IL-10 and Timp1 ⇑ |
| Leoci et al. | 8 | 1.05 | 5 min/2x/day for 3 weeks | canine prostate hyperplasia, ⇓ volume |
| Meyer-Wagner et al. | 15 | 5 | 45 min every 8 h, 3x/day for 21 days | ⇑ growth of bone marrow meschymal stem cells |
| Parate et al. | 15 | 2 | 1 application for 10 min | ⇑ Sox-9, COL II, and aggrecan. ⇑chondrogenesis via TRPC1 |
| Ross and Harrison | 5.1 (0-30) | 0.04 | 1 hr | Raw macrophage 5.1 Hz best for shutting down NFkB |
| Tang et al. | 15 | 1 | 0-8 6hr, 6 hr best ⇓ | IL‐1α induced IL‐6 expression in disc nucleus pulposus cells |
| Liu 2022 | 8 | 3.82 | 40 min/day, 5 days a week | osteoarthritis rat model |
Pyroptosis, 3.82 mT, 8 Hz PEMF, [4] and phosphatidyl inositols
Expanding on Liu 2022, [4] Low-grade inflammation is a key mediator of the pathogenesis of Osteoarthritis (OA). This study was interested in the relationship with the NLRP3/Caspase-1/GSDMD signaling pathway.
- After the proteolytic cleavage, the C-terminus remains in the cytosol while the N-terminal cleavage product interacts with negatively charged phosphatidylinositol 4-phosphate [PI(4)P] and phosphatidylinositol 4,5-bisphosphate [PI(4,5)P] on the inner leaflet of mammalian cell membrane where it oligomerizes to form a 16 subunits 10–14 nm pore.
- Water rushes in leading to cell swelling, lysis, and secretion of inflammatory cytokines
- GSDMD-N can also undergo cytoplasmic distribution and selectively bind to CL on inner and outer leaflets of intracellular bacterial membranes, or be secreted from pyroptotic cells through the pores into the extracellular milieu to target and kill extracellular bacteria.[16]
- Pyroptosis, which can now be defined as gasdermin-mediated necrotic cell death, acts as an immune defence against infection. Failure to express or cleave GSDMD can block pyroptosis and disrupt the secretion of IL-1β, and eventually unable to ablate the replicative niche of intracellular bacteria.
- Wikipedia authors mentioned downregulation of GSDMD in some cancers promoting proliferation due to the failure to inactivate ERK 1/2, STAT3 and PI3K/AKT pathways.[
- Sepsis and lethal septic shock can result from overactivation of pyroptosis.
Part of the below image came from a review on microdomains of phospholipids

Three-month-old Sprague-Dawley (SD) rats were divided into three groups (n = 6 per group): 1) OA 2) OA+PEMF 3) Control group (sham operation with placebo PEMF). Rats in the OA and OA+PEMF groups were subjected to bilateral anterior cruciate ligament transection and ovariectomy. The expression levels of NLRP3, Caspase-1, GSDMD, IL-1β, and MMP-13 were detected by qRT-PCR and Western blot. The results showed that PEMF alleviated the degree of inflammation and degeneration of cartilage in rats with OA, A decrease in cytokines IL-1β and matrix metallo protease 13 was observed. A decrease in the over-expression of NLRP3, Caspase-1, and GSDMD in the cartilage of the OA rats was observed. [4]
Efferocytosis at 75 Hz 1.5 mT[5]
Efferocytosis is the removal of dead apoptotic cells by phagocytosis. The process is generally considered anti inflammatory. Since these authors were LPS in the skin model, pyroptosis could be at play. Some H&E stained tissue sections are being presented as a visual to the notion that PEMF might just get charged toxins out of cervices in tissue via Faraday Induction. It should be noted that the Ouyang group had an entirely different explanation.
inflammation of the skin Figure 2
model in which air pouches with bacterial lipopoly saccharide (LPS) were used to induce inflammation in the skin. The mice were then treated by 75 Hz pulse wave form PEMF (1.5 mT, 75 Hz, 10% duty cycle). The synovitis scores were decreased by about a half as were the protein levels of IL-1β and TNF-α. p38 phosphorylation was decreased by PEMF and linked to decreases in mRNA for IL-1β and TNF-α. The disconnect being the conversion of the pro-enzyme IL-1β to the proteolytically cleaved active enzyme. [5]
PEMF reduces the degree of synovitis in the air pouch model.
- Figure 2 shows the procedure of mice treated with PEMF in air pouch model. Lipopoysccharide was involved to simulate an infection.
- PEMF reduced the levels of IL-1β and TNF-α cytokines in the lavage fluid.
- A small piece of the skin from the air pouches was sampled for H.E. staining, the purple part of H&E stains nuclei. No significant change in the number of nucleated cells after PEMF treatment. What is remarkable is how tight these spaces really are, bringing us to the Faraday Law of Induction hypothesis for how PEMF works.
Inflammation of the joint Figure 1
Synovitis is the inflammation of the synovial membrane and plays a part in arthritis. In Figure 1, note how tight the tissue seems to be. Do pulsating magnetic fields simply get charged particles moving thus assisting the body to heal itself?
After DMM surgery, one group of mice was treated with PEMF. After two weeks, all mice were sacrificed and the medial compartment of the knee was histologically evaluated. Histological evaluation: representative images of the medial compartment of the knee are revealed by H&E staining. Unlike the skin air pouch injections, IL-1β and TNF-α were not measurable. [5] The Ouyang study had much to say about inflammatory cytokines. These tissue images are shown to fuel the thought process that PEMF just causes charged particles to flow in confined spaces. The Ouyang group did not mention the “Faraday Law of Induction.” they did replicate the experimental 75 Hz PEMF treatment with the RAW macrophage cell line. We want our macrophage to be very diligent in clearing away dead cell debris that tends to be sources of inflammatory cytokines. This is a recycled image from the myoblast and ERK post describing the effect of 100 Hz PEMF on myoblast signaling.
In cultured myoblasts cells 100Hz and 1mT: ERK, JNK, p38 detour [6]
PEMF (100 Hz, 1 mT) promoted C2C12 myoblasts proliferation. The phosphorylation (activation) level of extracellular signal-regulated kinase (ERK) was significantly increased, while p38 MAPK and c-Jun N-terminal kinase (JNK) pathways were not affected. Pretreating the cells with the ERK kinase1/2 (MEK1/2) inhibitor U0126 inhibited the effects of PEMF.
ERK, JNK, p38 macrophage efferocytosis in macrophage Figure 4
Ouyang and coauthors stimulated RAW264.7 cells and bone marrow derived macrophage with LPS for 6 hours and then treated with PEMF for 30 minutes. [5]PEMF affects the phosphorylation of P38. The RAW264.7 cells (a) and BMDM cells (b) were stimulated with LPS for 6 hours and then treated with PEMF for 30 minutes. Phosphorylation (activation) of Three MAPK family members were analyzed by Western blots.
- ERK, growth factor, no change
- JNK stress, no change
- , p38, inlfammation, phosphorylation was weakened.
- messenger RNA from RAW264.7 cells revealed lower levels of inflammatory cytokines IL-1β and TNF-α
Faraday’s Law: mixing things up in inflamed regions?
We’ve got so many competing hypotheses as to how PEMF works, PEMF may sometimes seem like “pseudo science” even though the benefits are well established, particularly when it comes to inflammation. I’ve come to the concluding hypothesis that very low frequency PEMF generated from helmholtz coils simply stir up in toxic, inflamed regions so that the body can better detoxify and heal itself! As for this situation that in some cells PEMF causes ERK phosphorylation and in other cells it weakens p38 phosphorylation. Could it be that PEMF is simply accelerating the Brownian motion that would take growth hormones to and cytokines from to their respective receptors faster?
References
- Gaynor JS, Hagberg S, Gurfein BT. Veterinary applications of pulsed electromagnetic field therapy. Res Vet Sci. 2018 Aug;119:1-8. free paper
- Vincenzi F, Targa M, Corciulo C, Gessi S, Merighi S, Setti S, Cadossi R, Goldring MB, Borea PA, Varani K. Pulsed electromagnetic fields increased the anti-inflammatory effect of A₂A and A₃ adenosine receptors in human T/C-28a2 chondrocytes and hFOB 1.19 osteoblasts. PLoS One. 2013 May 31;8(5):e65561. PMC free article
- Ross CL, Ang DC, Almeida-Porada G. Targeting Mesenchymal Stromal Cells/Pericytes (MSCs) With Pulsed Electromagnetic Field (PEMF) Has the Potential to Treat Rheumatoid Arthritis. Front Immunol. 2019 Mar 4;10:266. PMC free article
- Liu J, Huang X, Zhou J, Li L, Xiao H, Qu M, Sun Z. Pulsed electromagnetic field alleviates synovitis and inhibits the NLRP3/Caspase-1/GSDMD signaling pathway in osteoarthritis rats. Electromagn Biol Med. 2022 Jan 2;41(1):101-107. PubMed
- Ouyang J, Zhang B, Kuang L, Yang P, Du X, Qi H, Su N, Jin M, Yang J, Xie Y, Tan Q, Chen H, Chen S, Jiang W, Liu M, Luo X, He M, Ni Z, Chen L. Pulsed Electromagnetic Field Inhibits Synovitis via Enhancing the Efferocytosis of Macrophages. Biomed Res Int. 2020 May 26;2020:4307385. PMC free paper
- Xu H, Zhang J, Lei Y, Han Z, Rong D, Yu Q, Zhao M, Tian J. Low frequency pulsed electromagnetic field promotes C2C12 myoblasts proliferation via activation of MAPK/ERK pathway. Biochem Biophys Res Commun. 2016 Oct 7;479(1):97-102. PubMed
Leave a Reply