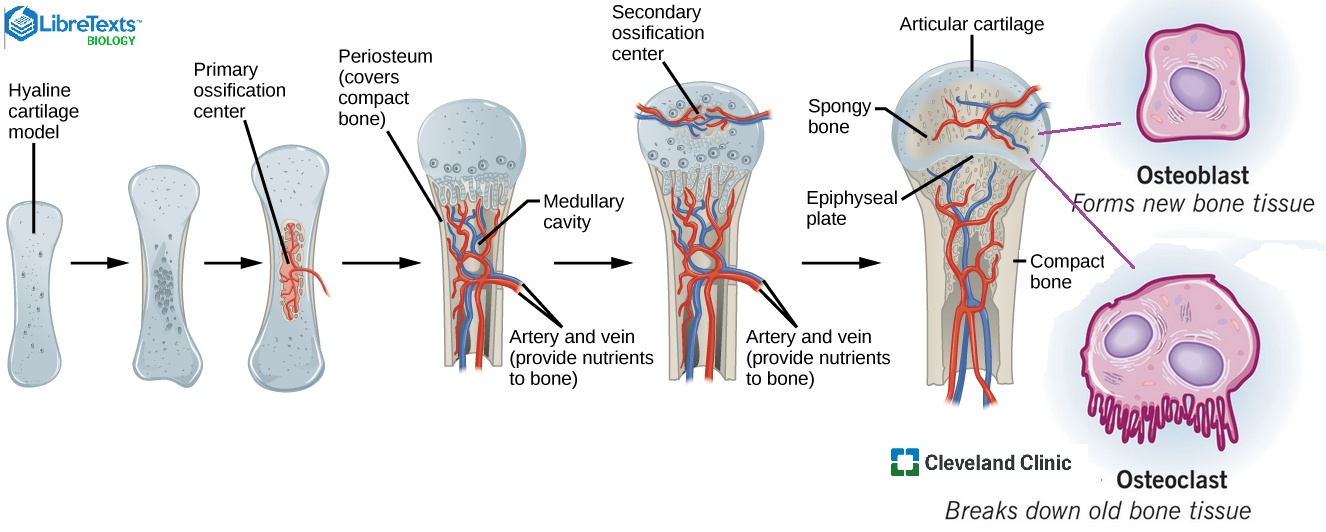
Both Magnawave and OAPP certification classes like to stress that PEMF helps the body heal itself by reducing inflammation. PEMF has been examined for 40+ years as a means of helping bones heal themselves. The Magnawave Semi-10 uses frequencies between 1-20 Hz depending on the setting. The magnetic field has a max of 4,700 G at the highest setting. As the setting increases the frequency decreases and the magnetic field increases. A table from a Wang 2024 review of studies dating back to 1983 is expanded upon. Many studies used higher frequency bursts at low frequency intervals. The Cho Lab of South Korea. This lab has examined in interplay between magnetic field intensity and frequency in the differentiation of bone resorbing osteoclasts: 7.5 Hz is pro osteoclast and 45 Hz is anti-osteoclast in cultured cells. The featured image from Libre Texts brings home the blood flow in bone growth. Libre Texts also emphasizes that bone remodeling, resorption and regrowth occur in regions of stress and muscle action. The Cleveland Clinic also reminds us of the importance of both processes for healthy bones. We need to remember that PEMF can improve blood flow in the general circulation and probably in bone.
The Semi-10 and Schuman resonances
Interesting Engineering The primary resonance is 7.83 Hz, with secondary and tertiary resonances a 14.1 and 20.3 Hz, respectively. Minor peaks were noted at 20.4 and 26.4 Hz with a spike at 50 Hz. The elecromagnetic fields originate from lightning strikes.

On the Semi 10, there is actually an underlying Schumann Resonance on every setting at 8 Hertz. The Schumann Resonance of earth varies greatly due to the amount of electromagnetic waves in the ionosphere. vary.
The Schumann Resonance in the 7-8Hz range can be more grounding, focusing more on overall wellness, mentality, etc. and the “bone ranges” on the Semi-10 tend to have more energy (gauss). For the Semi 10, the Hertz is between 1-20 depending on the setting and the gauss has a max of 4,700. Turning up the Semi-10 gets the magnetic fields deeper into the tissues.
The Wang 2024 table expanded
A wide range of high frequency micro pulses at lower frequency intervals worked well many decades ago. Back then the idea was to create pulsed magnetic fields to get charged particles and blood moving. They made note of the time to and from the burst magnetic field.
Wang A, Ma X, Bian J, Jiao Z, Zhu Q, Wang P, Zhao Y. Signalling pathways underlying pulsed electromagnetic fields in bone repair. Front Bioeng Biotechnol. 2024 Jan 24;12:1333566 PMC free article
The strategy was to copy the very basic table that only indicated “+” and “-” in the results column and fill in more details. The references have been replaced with links to free papers. Details on the fracture model have also been added. Interestingly, many models have combined PEMF and ceramics or even metal wires.
Table 1
| Fracture model | Treatment parameters | Results | References | Year |
|---|---|---|---|---|
| Rabbit Femur | Type: PEMF | -no change bone growth 22% increase in articular cartilage | Smith and Nagel (1983) | 1983 |
| Settings: repetitive pulse-72 Hz | ||||
| Duration: 12 h/day | ||||
| Sheep Tibia | Type: PEMF | -no change except increase in bone minerals | Law et al. (1985) | 1985 |
| Settings: 1.6 mT | ||||
| Duration: 24 h/day | ||||
| Horse Tibia, bone graph goal was to increase circulation to promote healing | Type: PEMF | PEMF-lower porosity percentage, more advanced corticalisation than untreated control grafts, | Kold et al. (1987) | 1987 |
| 30 msecs long @ 1.5 Hz with each pulse 250 positive of +2.4 mV peak and – 130 mv peak, 155x times in each burst. 0.7 mT | ||||
| Horse Metatarsal dorsal aspect of Mc3/Mt3 F may be repaired w/crews today. | Type: PEMF | Group 1 had less fibrous and woven bone. It was hard to determine which device was used in each group. | Sanders-Shamis et al. (1989) | 1989 |
| 21 pulses/burst, at 15 Hz, 20 G; and 30 msec pulse 100 pulses per burst, at 1.5 Hertz, 4 Gauss peak | ||||
| Duration: 8 h/day | ||||
| Rat Tibia | Type: PEMF | -no benefit healing non union fractures | Muhsin et al. (1991) | 1991 |
| Duration: 8 weeks | ||||
| Horse Metacarpus,diaphysial and metaphyseal holes | Type: PEMF | improve bone repair in bone marrow diaphyses and growth plate neck metaphyses | Canè et al. (1991) | 1991 |
| Settings: 28 G; 75 Hz | ||||
| Rat Mandible, injury demineralized bone matrix (DBM) | PEMF | >2x ⇑ in alkaline phosphatase & 3x ⇑ Ca2+ in the DBM graft-plus-PEMF group vs DBM-graft | Takano-Yamamoto et al. (1992) | 1992 |
| 10-msec-wide burst with 100 psec wide pulses, repeating at 15 Hz, mag field strength 1.5-1.8 G. | ||||
| Dog Mandible | Type: PEMF | prevented loss of ridge height after tooth extraction | Ortman et al. (1992) | 1992 |
| 100 Hz, a 10-msec-burst with 100 psec pulses, repeating at 15 Hz, 1.5-1.8 G. | ||||
| Rat Tibia | Type: PEMF | ⇑ callus formation | Sarker et al. (1993) | 1993 |
| 83Hz 20G peak Duration: 1 h/day | ||||
| Rat Spine | Type: PEMF 2.5mV | ⇑ callus formation | Guizzardi et al. (1994) | 1994 |
| Duration: 18 h/day | ||||
| Dog Lumbar spine | Type: PEMF | -no effect on the healing of the primary posterior spinal fusions | Kahanovitz et al. (1994) | 1994 |
| Settings: 1 G; 1.5 Hz | ||||
| Duration: 0.5–1 h/day | ||||
| Rabbit Humerus | Type: PEMF | direct current performed better in this study. | Yonemori et al. (1996) | 1996 |
| Settings: 2 G, 25 µs pulses at 10 Hz | ||||
| Duration: 12 h/day for 14 days | ||||
| Rabbits Femur | Type: PEMF | +0.2 mT better tan 0.3, 0.8 and control bone contact ratio and area | Matsumoto et al. (2000) | 2000 |
| 0.2, 0.3, 0.8mT for 8 h day pulse width (25 ms) (100 Hz) | ||||
| Duration: 4 h/day 1,2,4 weeks | ||||
| Rabbits Tibia | Type: PEMF | only stated low feq, improved callus formation | Fredericks et al. (2000) | 2000 |
| Duration: 1 h/day | ||||
| Dog Tibia | Type: PEMF | +enhancing effects of PEMF on callus formation and maturation in the late-phase of bone healing. | Inoue et al. (2002) | 2002 |
| Settings:1.5 Hz m 0 to -2 G in 230usec | ||||
| Duration: 4 h/day | ||||
| Rabbits Tibia, holes were drilled, filled w/ natural or synthetic apatite | Type: PEMF | PEMF benefits are early time points. Natural apatite is reabsorbed and replaced by natural bone. | Ottani et al. (2002) | 2002 |
| positive triange: 8 mT, 50 Hz, 2msec pause | ||||
| Duration: 0.5 h/day, 2-4 weeks | ||||
| Rabbits Tibia, titanium implant | Type: PEMF | -no influence in healing | Buzzá et al. (2003) | 2003 |
| Settings: pulse width 85 µs, ~11kHz | ||||
| Duration: 30 min/day | ||||
| Rabbits Tibia, metal device may have been involved, this was model of human leg lengthening | Type: PEMF | +PEMF improved stiffness and mechanical torque. PEMF increased callus formation by day 7 | Fredericks et al. (2003) | 2003 |
| 30 mSec bursts (33Hz) of asymmetric EM fields repeated 1.5 times a second (1.5 Hz) | ||||
| Duration: 1 h/day | ||||
| rat osteoblast cells | Type: static magnetic field | +Bone sialoprotein transcription, receptor tyrosine kinases involved, fibroblast growth factor-2 response element (FRE; pituitary-specific transcription factor-1 motif (Pit-1; | Shimizu et al. (2004) | 2004 |
| Settings: 300&800 G; | ||||
| ovariectomized Rat Tibia | electrical field | +electrical fields stimulate osteogenesis in ovariectomized female rats. | Lirani-Galvão et al. (2006) | 2006 |
| low-intensity pulsed (1.5 MHz, 30 mW/cm² | ||||
| Rabbits Tibia limb lengthening model | Type: PEMF | -no change in callus, torque, or stiffness in later stages. Better early callus formtion | Taylor et al. (2006) | 2004 |
| 30-ms bursts at1.5 Hz; during each pulse, the magnetic field rising from 0 to ~ 2 Gauss in 230ms and then 0 G in 30ms, | ||||
| Sheep Femur Cartilage repair with osteochondral autografts | Type: PEMF | PEMF ⇑ bone formation i⇑ graft stabilization. ⇓ bone resorption IL-1 | Benazzo et al. (2008) | 2008 |
| Settings: 1.5 mT; peak electric field 0.07 mV/cm 75 Hz | ||||
| Duration: 6 h/day | ||||
| Rat Tibia, titanium implant | Type: PEMF | day 10 ⇑ ossification index (p = 0.012). day 20 the osteotomies of the PEMF almost completely remodeled. ossification percentage ⇑ PEMF group (p = 0.018). | Grana et al. (2008) | 2008 |
| Settings: 72 mT; 30 Hz | ||||
| Duration: 1 h/day | ||||
| Rat Femur autogenous bone graph metal washers titanium screws | Type: static B field | magnetized washers ⇑ bone inte-gration 2. pmf ⇑ bone neoformation. | Puricelli et al. (2009) | 2009 |
| Settings: 41 G | ||||
| Rat Tibia, osteoporosis model | Type: PEMF | PEMF⇑ bone mineral density %TGF-b1 secretion ⇓ IL-6 expression | Shen and Zhao (2010) | 2010 |
| Settings: 8 G; 15 Hz | ||||
| Duration: 2 h/day | ||||
| Rabbits Femur fractured, magnetic and non magnetic metal implants. | static MF | NN fields ⇑ bone scores vs NS fields 2 magnets one femur | Aydin and Bezer (2011) | 2011 |
| Settings: 220–260 G | ||||
| Rabbit Tibia, more bone lengthening metal stuff | asymmetric pulse | -faster healing, no lon term difference | Taylor et al. (2012) | 2012 |
| 1.5 Hz30-msbursts MF r0-2 G in 230ms then 0 G in 30ms | ||||
| Duration: 20 days continuous | ||||
| ovariectomisedRat Tibia, look at cancellous and cortical bone | Type: PEMF | -no protection was seen. | van der Jagt et al. (2012) | 2012 |
| S1 G; 5-ms bursts with 5-μs pulses, repeat at 15 Hz | ||||
| Duration: 2 h/day | ||||
| Rat fractured Femur | Type: PEMF | both good alone but not in combination. | Atalay et al. (2015) | 2015 |
| Settings: 1.5 mT; 50 Hz Also PTX phosphodiesterase inhibitor | ||||
| Duration: 6 h/day for 30 days | ||||
| Rat Femur fracture with titanium nails | Type: PEMF | Note high frequency, enhanced early stage o f healing | Oltean-Dan et al. (2019) | 2019 |
| Settings: 6.65 mT; 27.12 MHz | ||||
| Duration: 10 min/day for 14 days | ||||
| Rabbit Tibia non union fracture, wires involved | PEMF vs combined MF | +Both are effective in promoting healing if used for at least six hours a day. | Fredericks et al. (2019) | 2019 |
| 4.5 ms bursts of 20 pulses @ 15 Hz. 0 ⇒ 1.6 mT 200 ms, ⇒0 T 25 ms. vs 76.6 Hz sine wave 20-40 mT superimposed with static 20 mT MF | ||||
| Duration: 6 h/day | ||||
| Rat delayed union fractur, posterolateral side of the left femur. K-wire knee to stabilize | Type: PEMF | PEMF helped early phase healing: ⇓ fibrous tissue area and ⇑ cartilage | Umiatin et al. (2021) | 2021 |
| Settings: 1.6 mT; 50 Hz | ||||
| Duration: 4 h/day for 5-28 days |
7.5 Hz, perhaps not that pro- osteoclast
The take home in looking at all of the graphs of tabular data from this publication is that the Schumann resonance frequency of 7.5 Hz is not that pro-osteoclastic when the intensity is ramped up. Most authors in the previous table used higher frequency bursts at lower frequency intervals.
Chang K, Chang WH, Wu ML, Shih C. Effects of different intensities of extremely low frequency pulsed electromagnetic fields on formation of osteoclast-like cells. Bioelectromagnetics. 2003 Sep;24(6):431-9. Sci-Hub free article
Magnetic fields induced within the solenoids were set for 1.3, 2.4, and 3.2 Gauss (0.13, 0.24, and 0.32 mT) rms and three different change rates of the magnetic flux density produced within the solenoids were set such to produce different induced electric field intensities as presented in data from tables as bar graphs. The calculated induced electric fields in the eight well slides at 7.5 Hz, with 0.8 ml medium (for a medium height of 0.8 cm and well width were 4.8, 8.7, and 12.2 mV/cm rms at the cell monolayer surface. Control cultures (i.e., 0 mV/cm) were grown separately.
- TNFα , according to Wikipedia authors, is thought to be primarily secreted by macrophages exposed to endotoxin. Other ells were reported to produce this cytokines. This cytokine was examined because of its importance in osteoclast differentiation. The cytokine was measured in the cell culture medium. Over the course of eight days there was only a doubling of TNFα at eight hours a day for eight days at 12.2 μV/cm. Other changes were minor.
- IL-1β is a product of the inflammasome and an inhibitor of bone formation. While some of these changes were “statistically” significant they were in the 10% range.
- Prostaglandin E2, PGE2, is a vasodilator in addition to other inflammatory processes. The authors did not see changes greater than about 50% and this was at day 8 with eight hours a day exposure at the highest magnetic field intensity.
Whether or not the changes are functionally large enough to matter, this mixed population of cells is not changing the production of cytokines that much.
7.5, 10, 15, 30, 45, 60, and 75 Hz, bone remodeling
This study used adipose derived stem cells (ASC) cultured in normal medium (NM) or osteogenic medium (OM). Magnetic fields compared were uniform but of different frequencies.
Kang KS, Hong JM, Kang JA, Rhie JW, Jeong YH, Cho DW. Regulation of osteogenic differentiation of human adipose-derived stem cells by controlling electromagnetic field conditions. Exp Mol Med. 2013 Jan 18;45(1):e6. PMC free article
The following is a summary of the figures:
- The authors provided color coded images of the magnetic fields generated by the cell culture incubator solenoids.
- In the radial direction the magnetic field was a flat 1 mT. In the axial direction the magnetic field peaked at 1 mT in the center and 0.5 mT at the ends.
- The frequency was held constant and normal growth medium (NM)) and osteogenic growth medium (OM) were compared for their ability to induce markers of differentiation in adipose derived stem cells. This figure was meant to define the system.
- Four different magnetic field intensities were evaluated for the six different frequencies: 1 mT was better at inducing alkaline phosphatase than 0.1, 2, and 3 mT over all frequencies. The frequencies 7,5, 60, and 75 Hz were essentially useless for inducing ALP. 30 Hz seemed to be optimal for inducing ALP expression.
- The bone transcription factor RUNX immunostaining was nil in the control and 7.5 Hz. Very good staining was seen at 30 and 45 Hz.
- OM (vs NM) was required for RUNX2 expression as well as collagin 1, osterix, and osteocalcin. 30 Hz was better than 45 Hz, and 7.5 Hz.
- Day 10 ALP activity and day 20 calcium content were greatest for 30 Hz compared to 45 Hz. There was a slight inhibitory action for 7.5 Hz compared to the control. OM, vs NM, was required to see this phenomenon.
The reason given for this 7.5 Hz to 30 Hz difference was a tipping point of 30 to 50Hz of Ca2+ flux through the membrane.
Koch CLMB M, Persson BRR, Salford LG, Eberhardt JL. Interaction between weak low frequency magnetic fields and cell membranes. Bioelectromagnetics. 2003;24:395–402. Sci-Hub free paper
In this case the cell membrane was plasma cell membranes with ATP driven Ca2+ pumps. The topping point was much closer to 24 Hz, the resonance frequency of Ca2+ and Mg2+. Following the reference trail was less than fruitful.
The seesaw of 7.5 and 45 Hz
This post has been struggling to emerge from notion that bone growth is all that matters in bone healing from a break. Bone loss is bad. This study used bone marrow stem cells.
Hong JM, Kang KS, Yi HG, Kim SY, Cho DW. Electromagnetically controllable osteoclast activity. Bone. 2014 May;62:99-107. Sci-Hub free article
This study involved bone marrow macrophage differentiating into osteoclast like cells. Please consult this possibly copyrighted miage from Research Gate.
- The 45 and 7,5 Hz solenoids produced similar magnetic field profiles ranging from about 0.8 to 1 mT.
- No interaction between PEMF and the macrophage colony stimulating factor (MCSF) concentrations was observed. 7.5 Hz increased the osteoclast formation by the RANKL differentiation factor.
- . BMMs were cultured with MCSF at day 0 or MCSF and RANKL (20 ng/ml) at days 2 and 4. The expression level of five osteoclastogenic markers: NFATc1, TRAP, CTSK,DC-STASMP, and the matric metallo protease 9. All markesr were significantly less than the control at 45 Hz and significantly more than the controls at 7.5 HZ.
- BMM were cultured on bone slices and MCSF and RNAKL to promote the formation of bone dissolving osteoclasts. Actin rings are part of this process. 7.5 Hz promoted the formation of actin rings while 45 Hz decreased it. The same could be said for the actin attachment protein beta integrin and the cytoskeletal protein tubulin.
- The influence of increasing concentrations of RANKL and 7.5 vs 45 Hz PEMF on MAPK members p38, JNK and ERK was examined.
- The deostification pits were much larger at 7.5 Hz and almost non existent t 35 Hz.
Conclusions
The exact settings of the Magnawave Semi-10 are proprietary. There is no patent information available. This is a company that has been successfully treating race horses and their bone issues for several decades. The research on wave forms from 20-30 years ago is plentiful. I am in the position of trusting the research even if the exact details are not given.

These days we exact frequencies for conditions. The more recent 7,.5 vs 45 Hz for osteoclasts probably has little bearing on complex wave forms that have been used for decades. Bone is also an organ with many different types of cells that include blood vessel cells. The good news is that we are always far lower in frequency than the deleterious EMF that have concerned society for decades.
Leave a Reply